- The paper introduces a novel Hodge-theoretic framework that decomposes brain network flows into dissipative and persistent components for counterfactual causal analysis.
- It quantitatively assesses network resilience using energy perturbation methods, simulating interventions such as anterior temporal lobectomy in temporal lobe epilepsy.
- The study demonstrates that pathological amplifications and compensatory shifts in temporal-limbic circuits provide crucial insights into therapeutic efficacy.
Counterfactual Causal Analysis of Brain Network Dynamics via Hodge Theory
Overview
The paper presents a rigorous framework for the counterfactual analysis of directed brain network dynamics, leveraging Hodge theory and energy perturbation methods to systematically model both pathological disruptions and therapeutic interventions. Unlike traditional acyclic causality methods (e.g., Granger causality, SEM, DCM), this approach decomposes network communication into dissipative and persistent (harmonic) components, enabling a principled examination of how causal organization adapts under hypothetical manipulations. The framework provides quantification of network resilience, compensation, and control through energetically informed reconfigurations and is demonstrated in applications to temporal lobe epilepsy (TLE).
Methodological Framework
Data Acquisition and Preprocessing
The underlying empirical basis derives from resting-state fMRI datasets of 400 Human Connectome Project subjects, preprocessed to mitigate motion artifacts and parcellated via the AAL atlas into 116 regions. Temporal dynamics of directed connectivity were estimated using time-lagged Pearson correlations in sliding windows matching the minimum hemodynamic response timescale. Edge flows between regions were robustly extracted by thresholding and employing higher-order motifs through simplicial complexes to preserve topological consistency across scales (Figure 1).
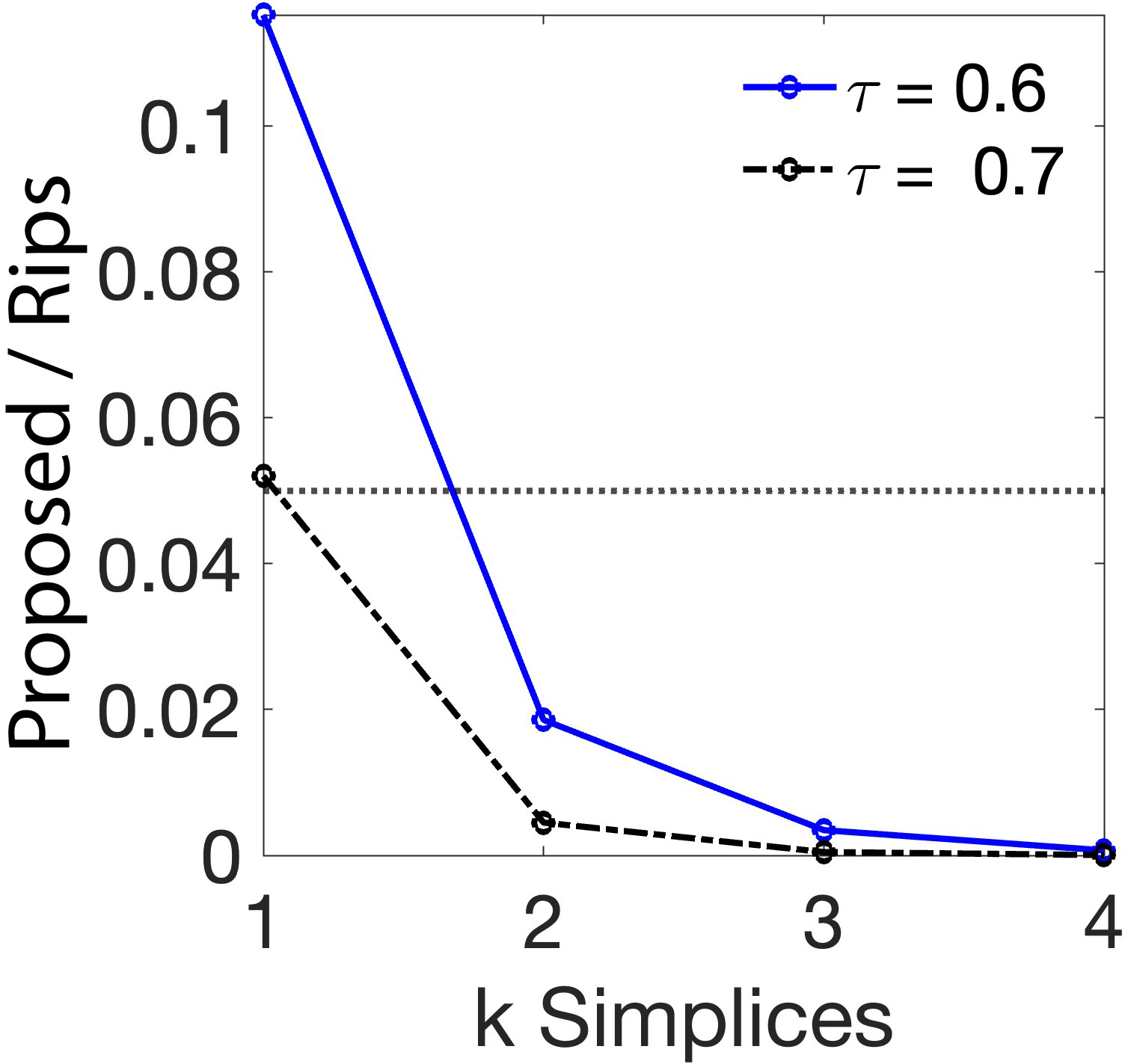
Figure 1: Left—Average fraction of simplices retained by the spatial scaffold relative to the maximal Rips complex; Right—Average time-lagged correlation across subjects and time points with maximal r=0.034, indicating no significant connections.
Hodge-Theoretic Decomposition and Energy Landscape
Directed network flows are formalized as time-varying edge flows, with causal propagation governed by minimization of Dirichlet potential energy associated with the 1-Hodge Laplacian. This operator acts on edges, encoding both divergence (source–sink) and rotationality (cyclic motifs). The Dirichlet energy,
E(X)=21⟨X,L1X⟩=21∥B1X∥22+21∥B2⊤X∥22,
provides a variational principle for information transport, with dynamics evolving toward minimum-energy steady states. The harmonic flow XH is singled out by L1XH=0, representing the persistent, non-dissipative backbone of communication that survives after all transient modes decay.
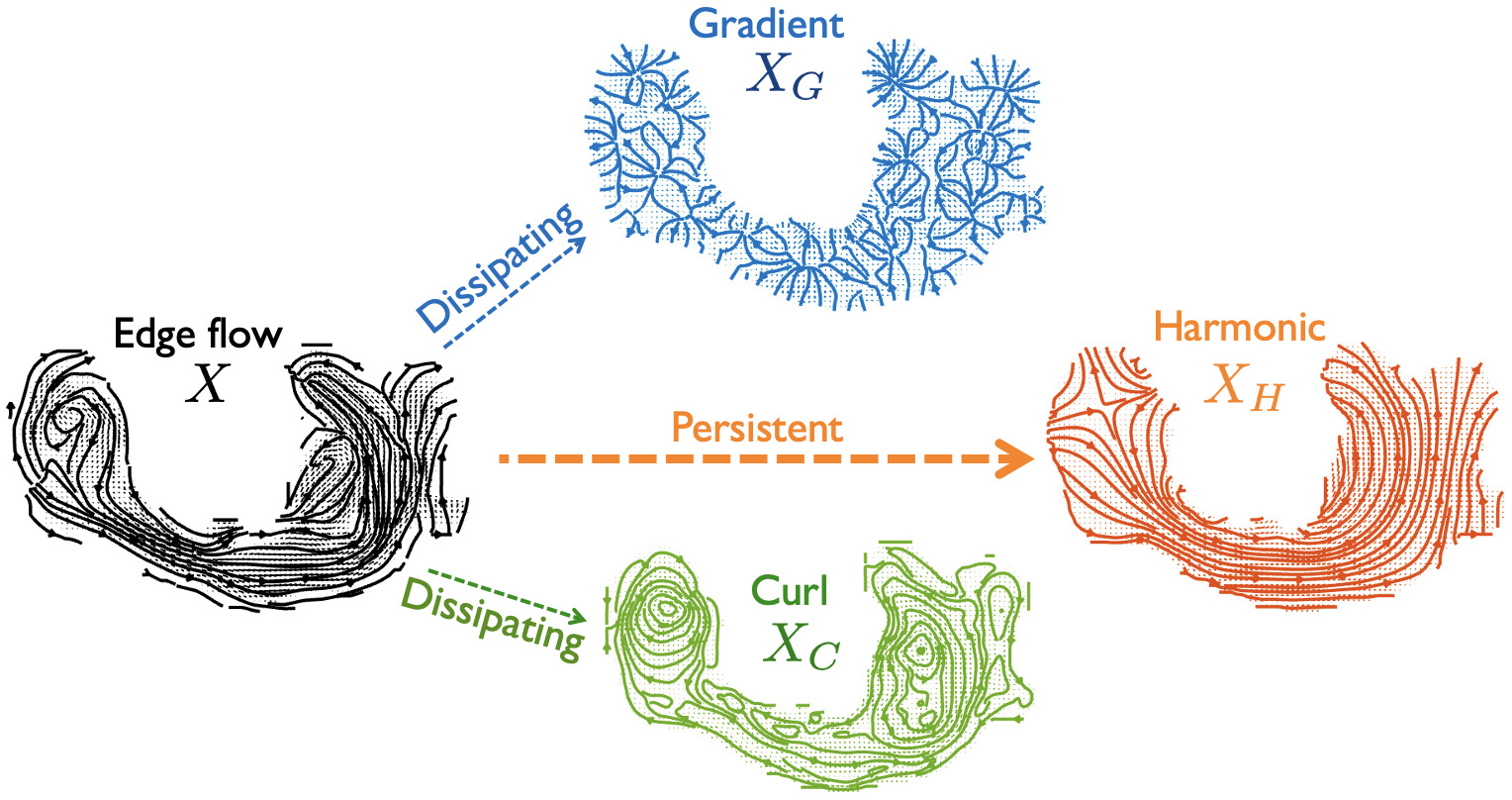
Figure 2: Minimization of Dirichlet potential energy in edge flows converges toward the harmonic flow XH; residuals capture dissipative (gradient) and cyclic (curl) components.
Counterfactual Operators and Stability Metrics
Counterfactual operators C act linearly on edge flows, simulating interventions by scaling, deleting, or modifying connection weights. The resultant energy change,
ΔE=E(CX)−E(X),
quantifies network fragility and the efficacy of compensatory routing. For harmonic flows, ΔE=0 under any linear counterfactual, emphasizing topologically invariant but energetically conserved reorganization.
Applications to Temporal Lobe Epilepsy
Counterfactual interventions were instantiated to probe functional resilience in TLE.
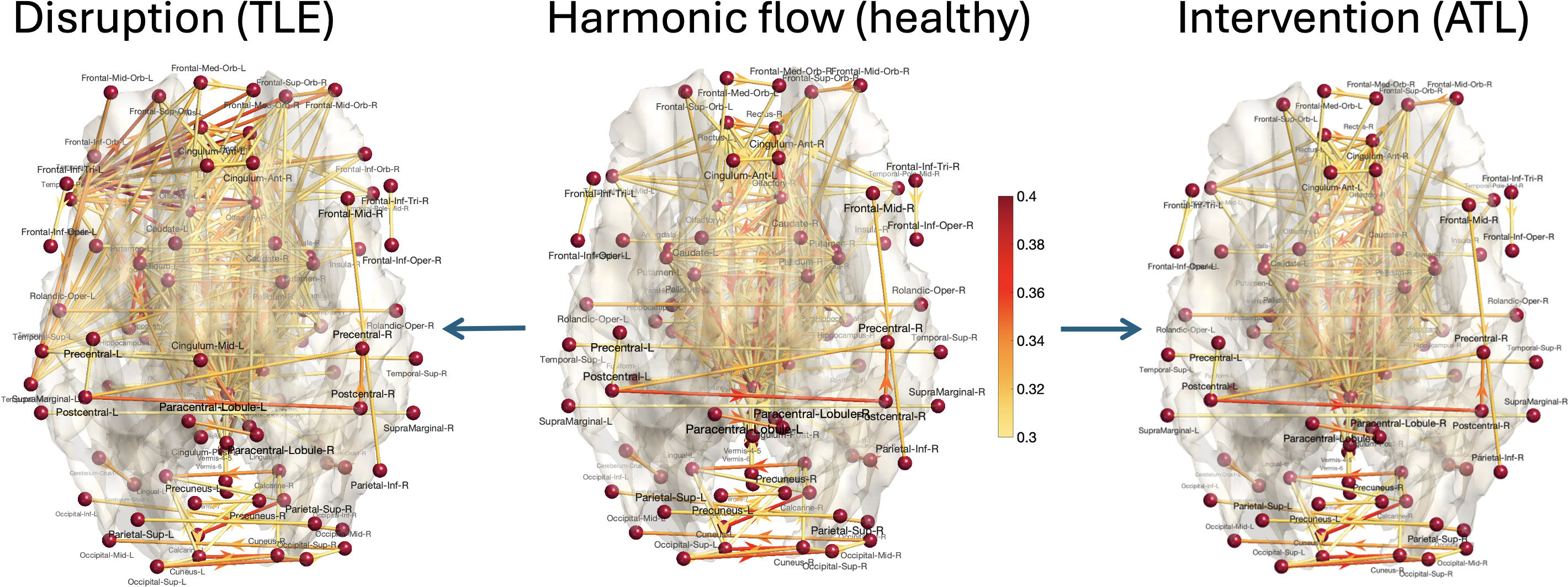
Figure 3: Middle—Dominant average harmonic flows (XH) for healthy subjects, highlighting recurrent interhemispheric and homotopic organization; Left—Pathological perturbation of limbic feedback loops in TLE shifts dominance to limbic and subcortical circuits; Right—Simulated left anterior temporal lobectomy preserves global topology via contralateral/midline compensation.
Disease Model Perturbation
Amplification of temporal-limbic feedback loops by 30% via harmonic counterfactuals in pathological TLE yields a shift in dominant persistent flows away from interhemispheric sensory circuits toward limbic and subcortical pathways (e.g., Olfactory→Amygdala, AmygdalaE(X)=21⟨X,L1X⟩=21∥B1X∥22+21∥B2⊤X∥22,0Olfactory). This mirrors empirical findings in TLE patient populations, where seizure propagation follows hippocampal–amygdalar circuits and engages cerebellar and pallidal structures.
Therapeutic Simulation: Anterior Temporal Lobectomy
Simulating functional ablation of the left temporal pole (ATL) attenuated limbic flows by 70%. In healthy controls, this showed minimal impact on persistent harmonic topology, suggesting robust compensatory stabilization via contralateral and midline circuits. In TLE, the same perturbation produced extensive redistribution toward limbic dominance, evidencing decreased compensatory capacity in pathological networks.
Implications and Future Directions
The formalism advances the theoretical foundation for causal inference in brain networks, supporting the systematic simulation of interventions (virtual lesioning, neuromodulation, adaptive reorganization) that are not directly attainable in vivo. Persistent harmonic counterfactuals furnish robust metrics for network resilience and redundancy, guiding the design of network-control paradigms in neurotherapeutics. Practically, the approach enables energetic, topological, and causal quantification of intervention outcomes, with immediate ramifications for presurgical planning and neuromodulation strategies.
Possible future directions include:
- Extension to multimodal imaging (e.g., EEG–fMRI) for validating counterfactual dynamics across data types.
- Incorporation of nonlinear or state-dependent counterfactual operators for modeling complex feedback loops.
- Theoretical investigation of energetic compensation limits and their relationship with functional recovery.
Conclusion
The paper introduces a mathematically rigorous framework for counterfactual analysis of brain networks, centered on Hodge theory and Dirichlet energy minimization. By decomposing flows into persistent harmonic components, it exposes the backbone of causal organization and offers principled tools for quantifying resilience, compensation, and stability under both pathological and therapeutic interventions. The method is empirically validated on temporal lobe epilepsy, elucidating differential reorganizational capacities in healthy and diseased states, and opens new avenues for energy-informed causal modeling and control in complex brain systems.


