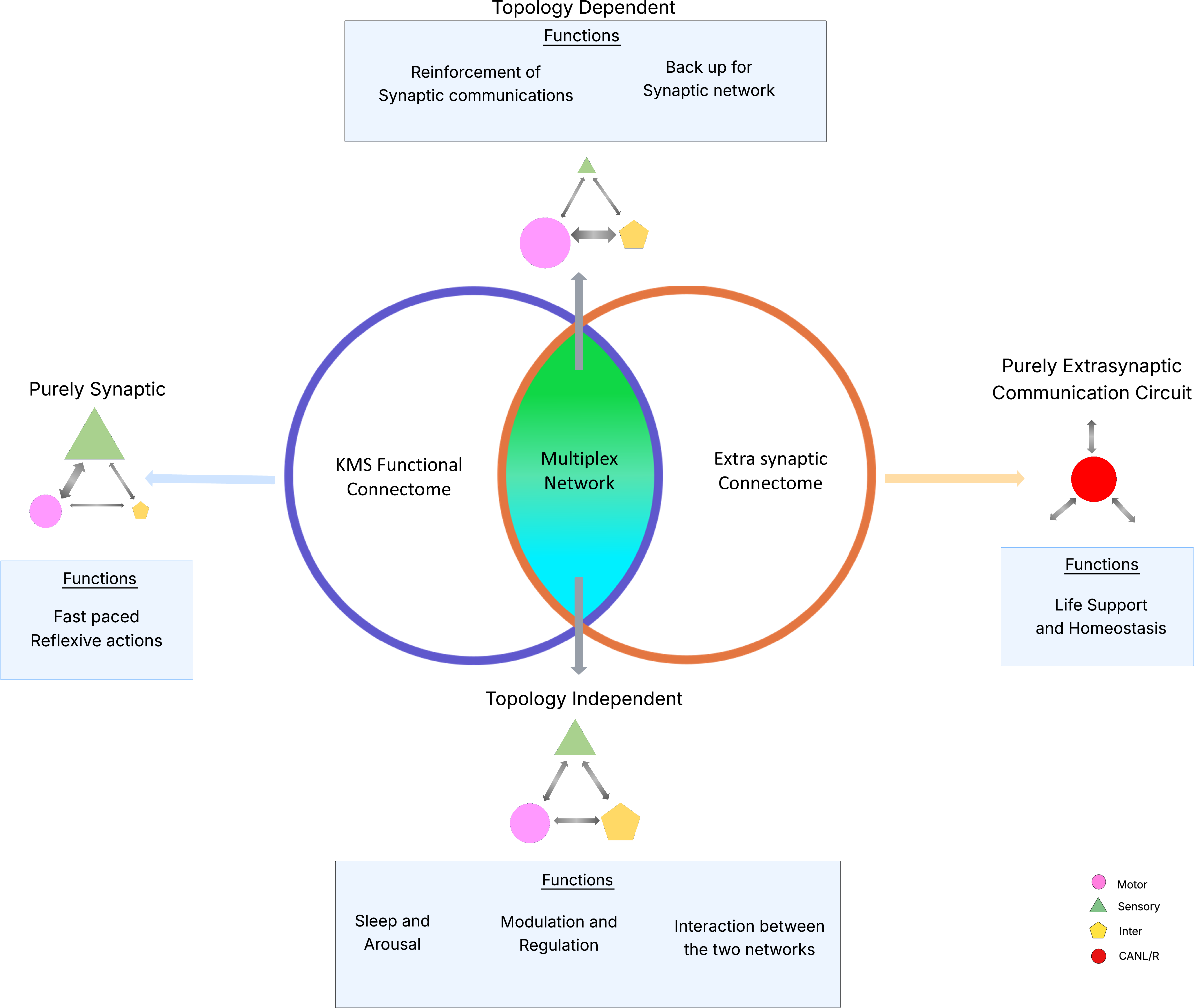
- The paper introduces a Kubo-Martin-Schwinger equilibrium framework to generate a probabilistic functional connectome that integrates synaptic and extrasynaptic signaling.
- The analysis reveals four distinct regimes—topology-dependent, topology-resilient, purely extrasynaptic, and purely synaptic—each linked to specialized neural functions like motor control and global modulation.
- Entropy and centrality measures validate that combining molecular and structural connectomics refines our understanding of network robustness and behavior.
Introduction and Theoretical Basis
This work analyzes the integration of synaptic and extrasynaptic (neuropeptidergic) signaling using the complete connectomes of Caenorhabditis elegans. The central methodological innovation is the application of the Kubo-Martin-Schwinger (KMS) equilibrium formalism to the large-scale connectome, treating the network as a system at thermal equilibrium and generating a probabilistic, structure-informed functional connectome (SIFC) that quantifies the expected influence of one neuron on another when all network motifs (including loops and multi-step pathways) are considered. This functional reference is then systematically compared to the molecularly defined extrasynaptic (neuropeptide-receptor) connectome, enabling the classification of signaling regimes according to the degree and nature of coupling between the two communication layers.
KMS-Derived Functional Connectivity
Conventional connectomics describes direct synaptic wiring but does not resolve how recurrent, multi-step circuits modulate information propagation. The KMS construction applies statistical physics to the directed graph, using an "inverse temperature" β to tune path lengths: high β weights short direct connections, lower β admits long-range propagation via indirect, recurrent, and feedback loops. The analytic computation converges for β>βc determined by the adjacency spectral radius, with the "critical" regime maximizing entropy of the mixed emittance distribution. All analyses use β=5.103 (determined empirically) to yield maximally diffusive, yet stable, patterns.
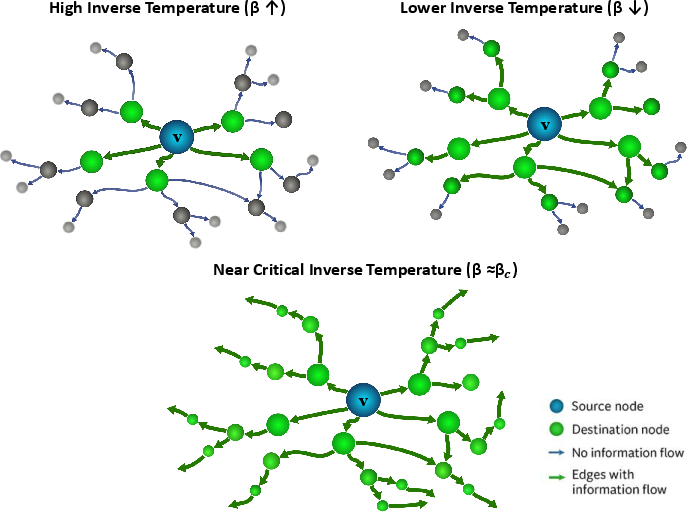
Figure 1: KMS-derived emittance profiles reveal the β-dependent reorganization of information flow, transitioning from local to distributed connectivity near βc.
The resulting SIFC captures the full distribution of probabilistic influences over the network and provides a benchmark for interpreting whether extrasynaptic connections reinforce, complement, or diverge from synaptic architecture.
Multiplex Organization of Communication Regimes
The SIFC and extrasynaptic network are integrated in a multiplex, two-layer representation, enabling the delineation of four signaling regimes based strictly on the overlap and statistical significance (null model analysis) of edges in SIFC and the extrasynaptic connectome. This classification is agnostic to cell type and is a direct function of network structure and molecular signaling:
Topology-Dependent Regime: Synaptic Motor Circuit Reinforcement
The topology-dependent regime contains 5,254 connections, primarily mapping onto motor neurons essential for locomotion (e.g., DB, DA, VA, AS classes). Degree and centrality analyses highlight strong in-degree convergence onto motor hubs (DB05, DA07, VA10, AS08), with some nodes (VB07, AVBL, AVBR) acting as key broadcasters. The extrasynaptic component in this regime is structurally aligned with SIFC, indicating that neuropeptide signaling reinforces and stabilizes existing synaptic circuits.
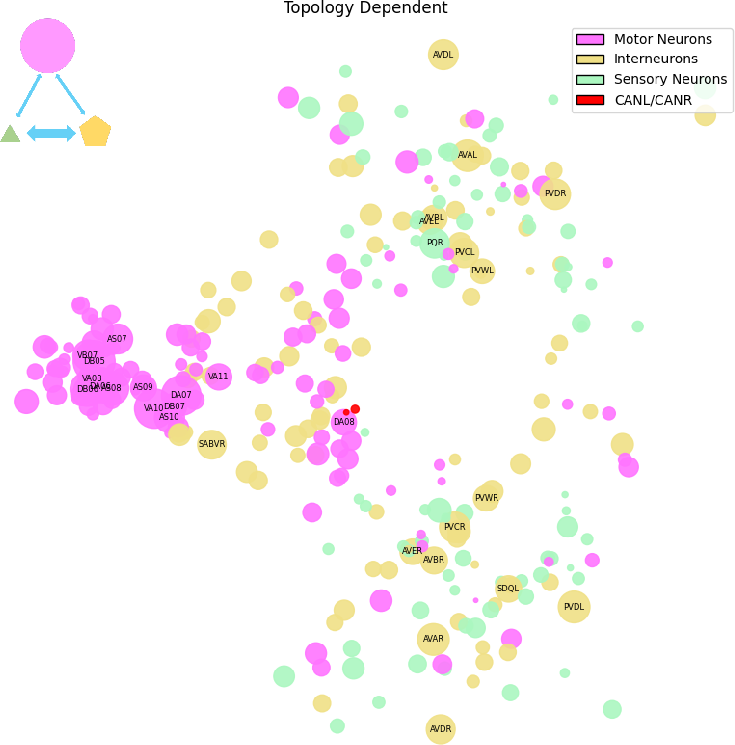
Figure 3: Topology-dependent regime overlays extrasynaptic edges onto synaptic communication hubs, predominantly reinforcing motor circuits.
This structure enhances communicational robustness to wiring perturbations, providing functional redundancy and error protection for critical behaviors.
Topology-Resilient Regime: Global Modulation Decoupled from Synaptic Topology
Comprising 42,022 edges, the topology-resilient regime remains stable after randomization of synaptic wiring and exhibits a distributed, symmetric structure dominated by interneurons involved in behavioral state regulation (e.g., AVK, PVQ, RIM, DVA, RIB). The balanced in/out-degree distribution facilitates broad, network-wide modulation and coordination, with prominent integration of environmental stimuli (e.g., URX for oxygen sensing).
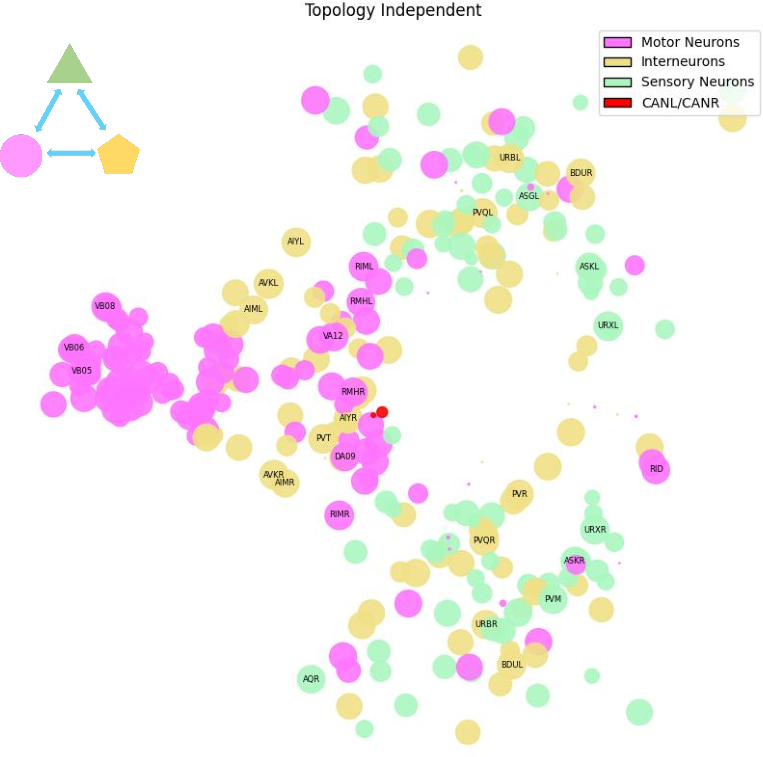
Figure 4: The topology-resilient regime forms a highly interconnected and symmetric extrasynaptic network among interneuronal populations.
This regime's wiring independence supports volume transmission and state-dependent regulation across the nervous system, unconstrained by fast, point-to-point connectivity.
Purely Extrasynaptic Regime: Survival and Homeostasis
This regime, with 6,759 edges, exclusively serves neurons with minimal synaptic wiring but maximal extrasynaptic connectivity—CANL/R, M1, M4, I1—which are individually essential for viability, feeding, and metabolic regulation. These pathways are not mirrored by SIFC or its null-model analogues, confirming that vital homeostatic and organismal maintenance is subserved by diffusive neuromodulation rather than classic synaptic dynamics.
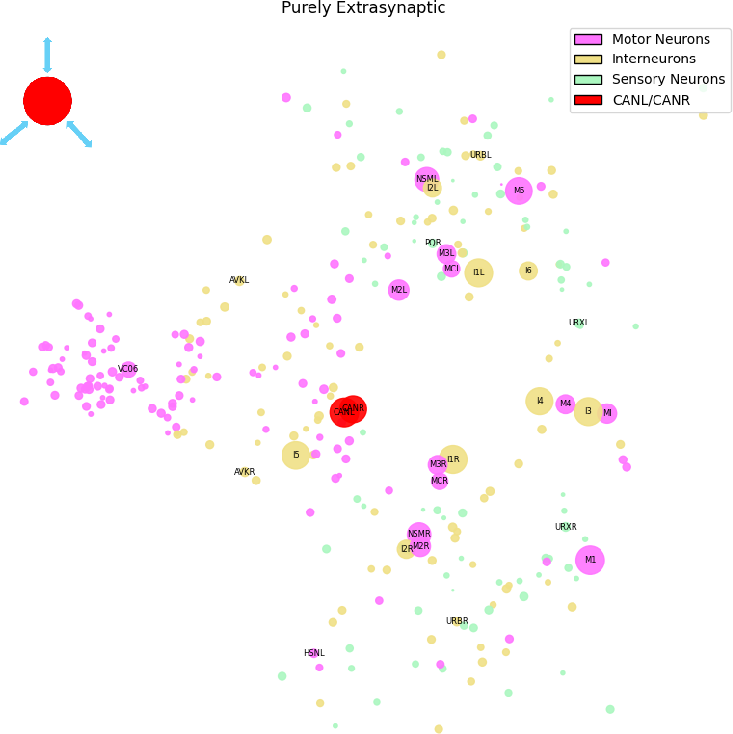
Figure 5: Purely extrasynaptic regime supports core survival and homeostatic behaviors via high-degree, extrasynaptic hubs.
The structure-functional dissociation underscores the necessity of parallel, non-synaptic communication channels for essential physiological processes.
Purely Synaptic Regime: Rapid Sensorimotor Transmission
The purely synaptic regime encompasses 30,950 connections (2,649 statistically significant), primarily among RME, PHC, AS10, and FLP neurons governing fast sensorimotor and reflexive responses. These pathways are characterized by absent extrasynaptic support, emphasizing exclusive reliance on rapid, classical synaptic transmission.
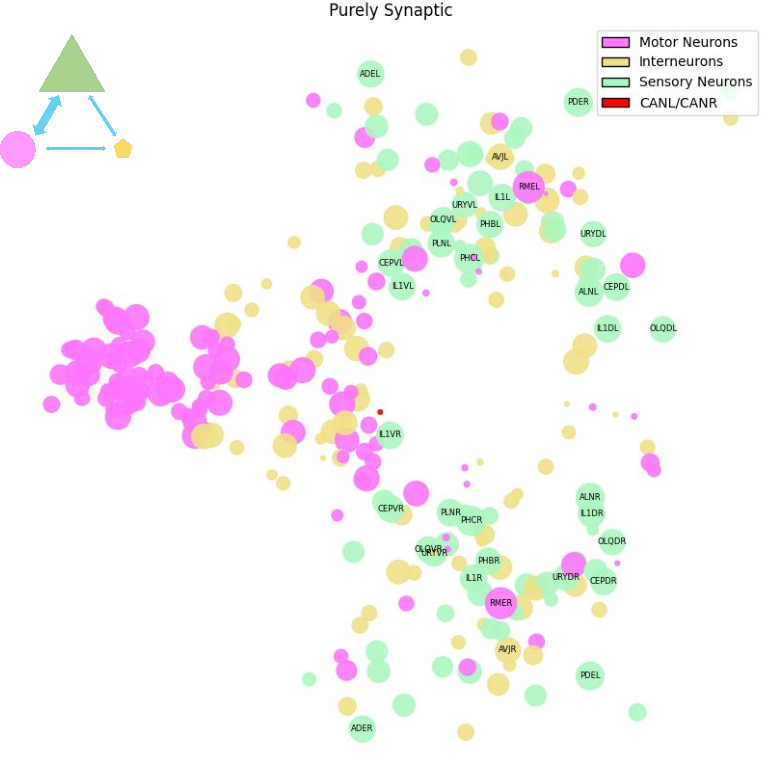
Figure 6: Purely synaptic regime is specialized for high-speed, low-latency sensorimotor responses.
Such separation ensures unimpeded signal fidelity where temporal precision is paramount and modulatory transmission would introduce undesirable delays.
Entropic Characterization and Centrality Analysis
Entropy analysis demonstrates that the chosen β yields maximally informative and distributed functional organization. Centrality measures within sub-graphs support the regime-specific specialization described above. Notably, degree and betweenness centrality patterns confirm integration roles for motor neurons in topology-dependent subgraphs, balanced distribution for interneuronal modulators, and exclusively high-degree CAN neurons in the purely extrasynaptic context.
Implications and Prospective Directions
This multiplex decomposition demonstrates that extrasynaptic signaling constitutes a structured, functionally specialized architecture fundamentally distinct from—and not subordinate to—synaptic wiring. The identification of a topology-resilient, modulatory layer, and a purely extrasynaptic regime uniquely associated with survival functions, underscores the necessity of integrating molecular and topological modalities for a comprehensive understanding of network computation and behavioral output.
The approach lays the foundation for advances in multilayer/heterogeneous network modeling, where chemical and electrical synapses, as well as multiple classes of modulatory connections, may be parametrized distinctly. Expansion to other organisms will become feasible as complete connectomic and neuromodulatory maps become available. Methodologically, the equilibrium approach provides a complementary alternative to dynamical models, extracting the latent computational blueprint for information flow architecture.
Conclusion
This study establishes a rigorous functional reference for analyzing multiplex neural communication and demonstrates, in the fully mapped C. elegans nervous system, clear division of labor among synaptic and extrasynaptic regimes. The findings refute the notion of extrasynaptic signaling as merely secondary to synaptic transmission, revealing role-optimized, non-overlapping sub-architectures specialized for speed, global modulation, robustness, and organismal survival. This multiplex perspective provides a template for systematic, principled integration of structural and molecular connectomics in neural systems.
References:
"Thermodynamic connectivity reveals functional specialization and multiplex organization of extrasynaptic signaling" (2604.02057).