- The paper demonstrates that combining dilatation and distensibility data with neural operator frameworks yields sub-10% prediction errors for mechanobiological insult mapping.
- UNet architecture outperforms other models by accurately localizing heterogeneous insults and minimizing spurious predictions in TAA imaging.
- The study underlines the need for advanced imaging protocols to capture full-field mechanical properties and enhance personalized TAA risk stratification.
Neural Operator-Based Identification of Mechanobiological Insults in Thoracic Aortic Aneurysm
Introduction
This study addresses the challenge of inferring the underlying mechanobiological determinants of thoracic aortic aneurysm (TAA) from noninvasive imaging-derived data. The clinical management of TAA is currently predicated on geometric metrics, primarily maximum aortic diameter, which are insufficient for risk stratification due to the heterogeneity of underlying pathophysiology. The paper proposes a machine learning framework leveraging operator-based neural networks to map full-field measurements of aortic dilatation and distensibility to spatially heterogeneous, multi-contributor insult profiles—specifically, compromised elastic fiber integrity and dysfunctional mechanosensing. The approach is validated on a synthetic dataset generated via finite element (FE) simulations parameterized to murine models of TAA, with a systematic comparison of DeepONet, UNet, and Laplace Neural Operator (LNO) architectures.
The synthetic dataset is constructed by superimposing spatially heterogeneous insults to elastic fiber integrity and mechanosensing, parameterized via Gaussian random fields (GRFs) over the circumferential and axial coordinates of the aortic wall. Each insult profile is mapped to FE simulations of aortic growth and remodeling, yielding steady-state geometries under physiological loading. From these, two scalar fields are extracted: local dilatation (normalized inner radius) and distensibility (normalized difference in radius between systole and diastole). The dataset comprises 500 pairs of dilatation-distensibility maps, each associated with the ground-truth spatial distribution of the two insult contributors.
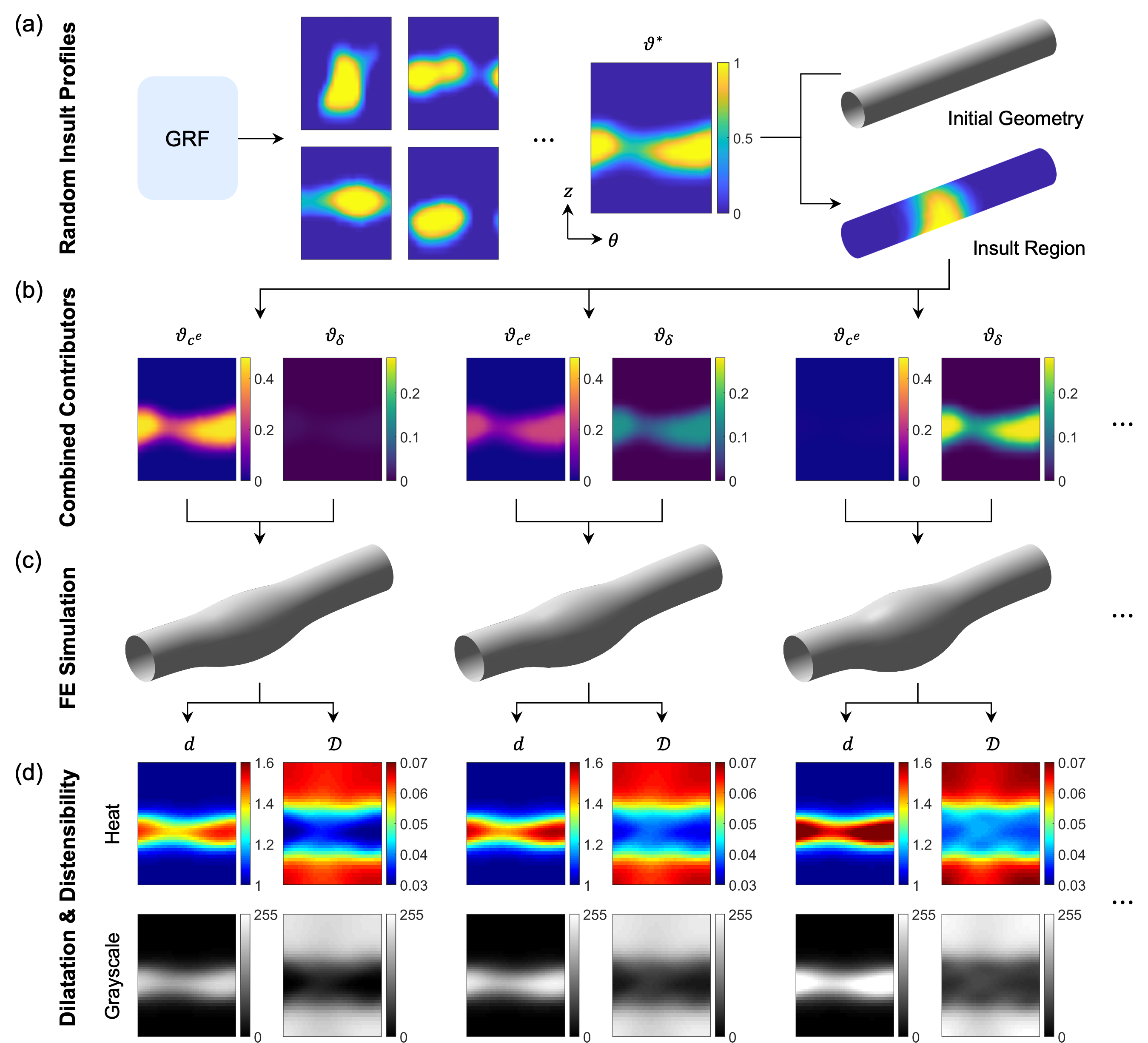
Figure 1: Synthetic data generation pipeline, from random insult field generation to FE simulation and extraction of dilatation and distensibility maps.
The problem is cast as a supervised learning task: given a pair of 2D maps (dilatation and distensibility), predict the underlying spatial fields of elastic fiber integrity loss and mechanosensing dysfunction. The study further investigates the impact of input data format (grayscale vs. heat map) and the inclusion/exclusion of distensibility information, reflecting the limitations of current clinical imaging protocols.
Neural Operator Architectures
Four neural operator architectures are evaluated:
- DeepONet (CNN and FNN branches): The branch network encodes the input maps (either as images via CNN or as vectors via FNN), while the trunk network encodes spatial coordinates. The output is the predicted insult field at each spatial location.
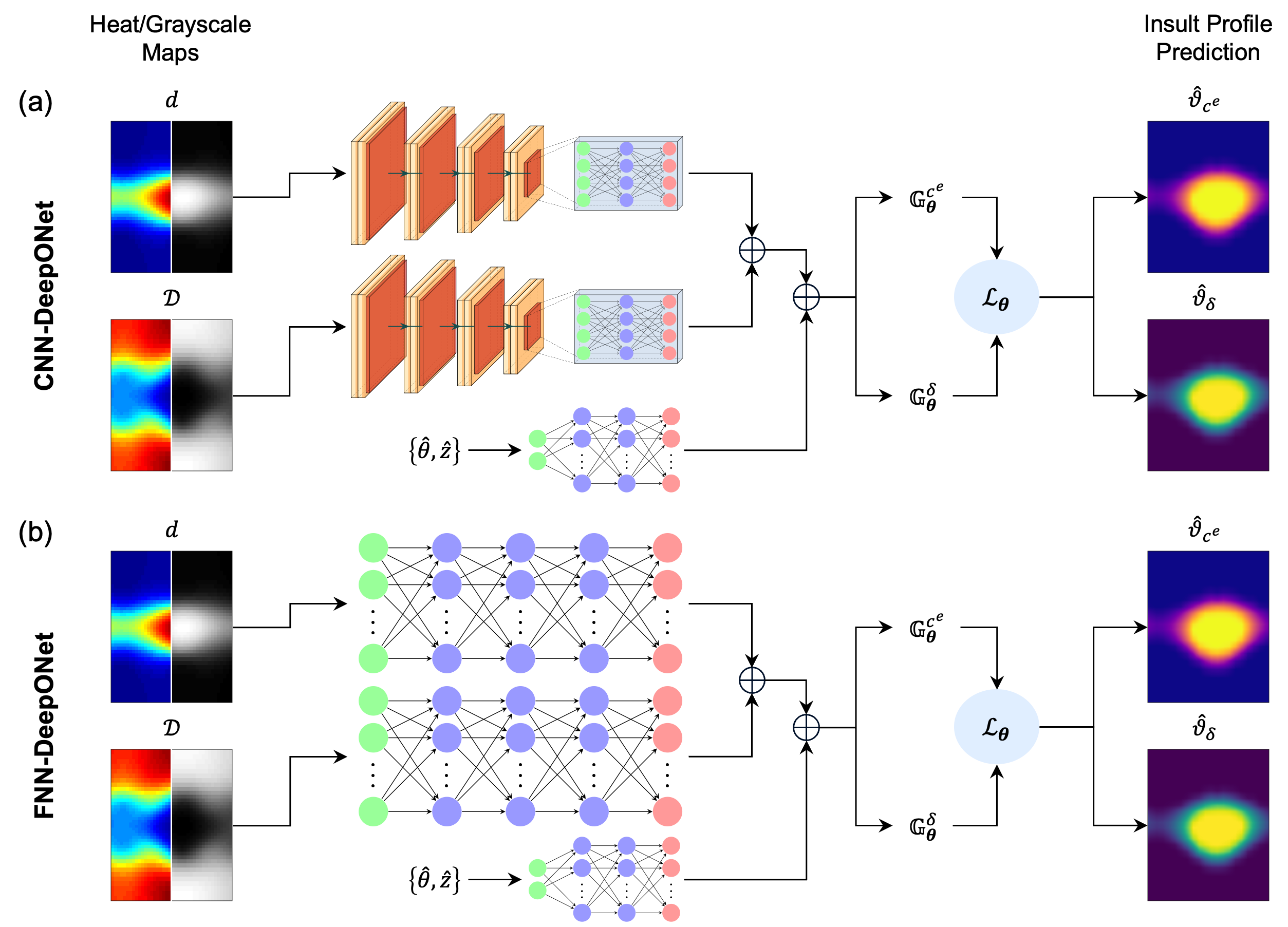
Figure 2: Schematic of DeepONet architectures with CNN and FNN branches for encoding input maps.
- UNet: A fully convolutional encoder-decoder with skip connections, mapping input maps directly to output insult fields. The architecture is well-suited for capturing multi-scale spatial correlations.
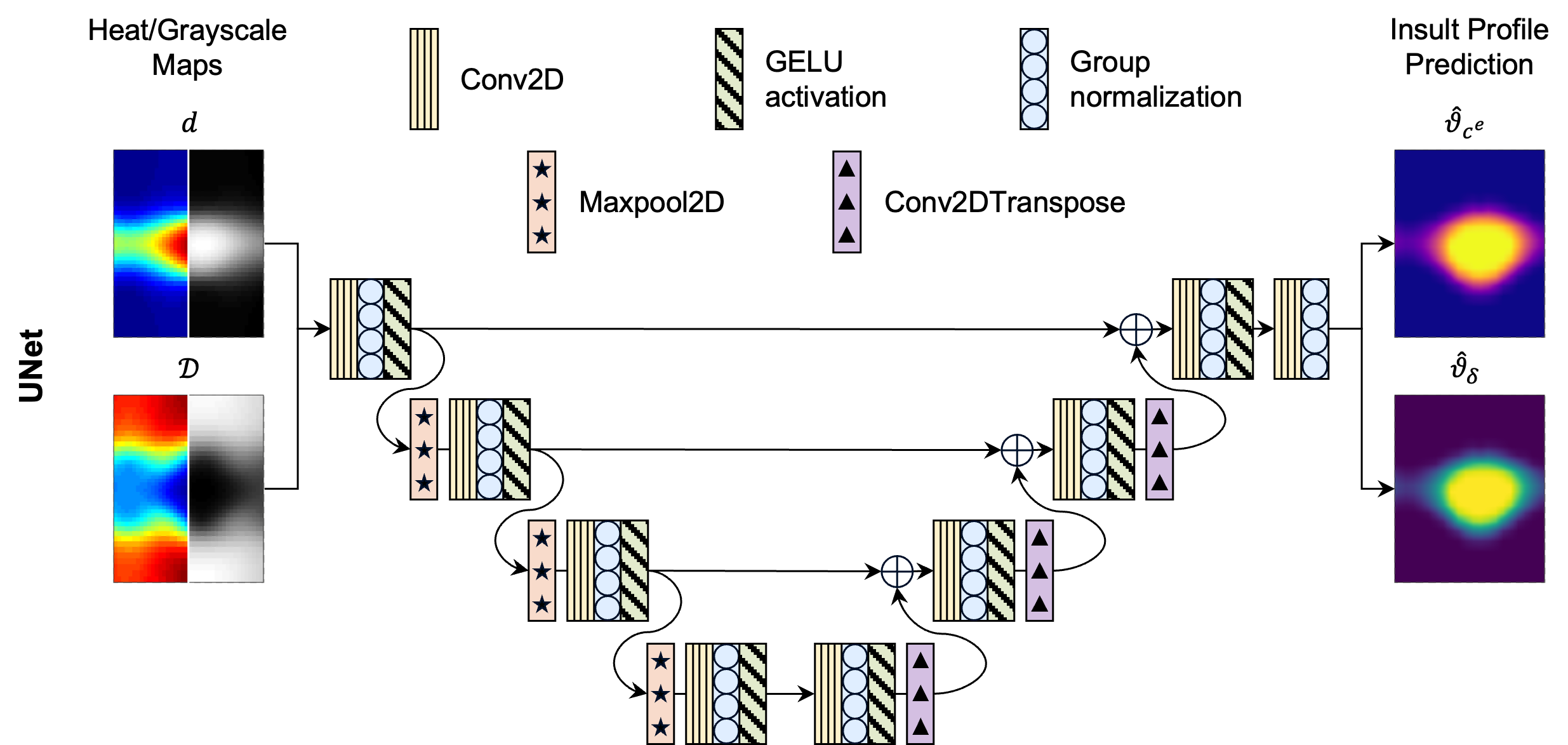
Figure 3: Schematic of the UNet architecture for mapping input maps to insult fields.
- Laplace Neural Operator (LNO): Operator learning in the Laplace domain, leveraging pole-residue decompositions to capture both steady-state and transient system responses.
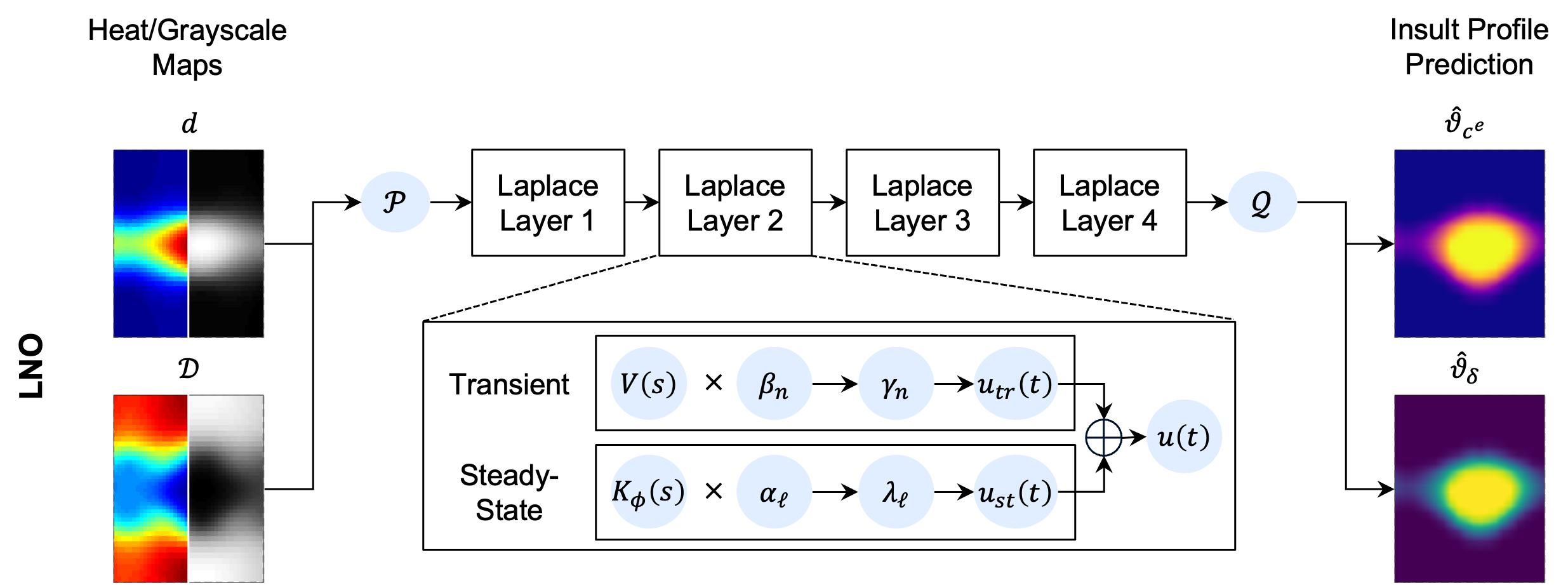
Figure 4: Schematic of the LNO architecture, illustrating the Laplace domain mapping and projection back to the spatial domain.
All models are trained with a relative L2 loss between predicted and ground-truth insult fields, with hyperparameters selected via grid search and convergence monitoring.
Results
All architectures achieve sub-10% relative L2 error in predicting both insult contributors when trained on both dilatation and distensibility maps. The UNet consistently outperforms other models, achieving the lowest errors for both elastic fiber integrity and mechanosensing, regardless of input data format.
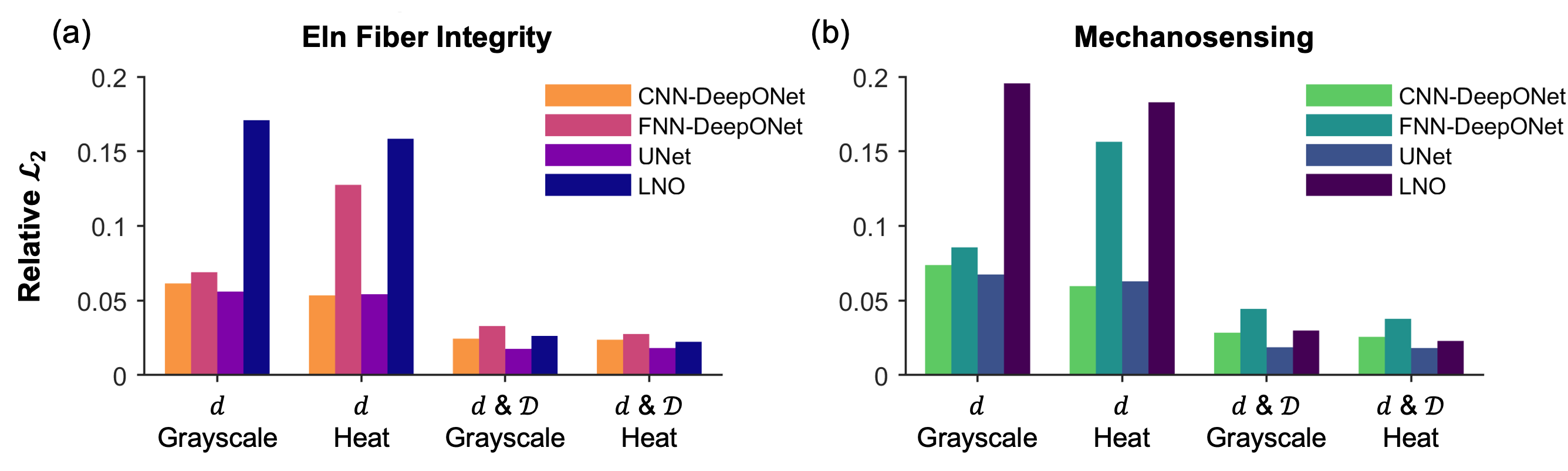
Figure 5: Relative L2 errors for all architectures and input data formats, for both insult contributors.
When trained on dilatation alone (mimicking current clinical practice), prediction errors increase by a factor of approximately two across all models. The inclusion of distensibility is thus critical for accurate inference of underlying pathophysiology, particularly in cases where different insult combinations yield similar geometric dilatation but distinct mechanical properties.
Qualitative Analysis of Predictions
Representative examples demonstrate that UNet and CNN-DeepONet architectures localize the insult region with high fidelity, while FNN-DeepONet and LNO exhibit greater errors, especially in dilatation-only scenarios. Errors are concentrated in the insult region, with UNet and LNO minimizing spurious predictions in unaffected regions.

Figure 6: Predictions from all four architectures for an elastic fiber integrity-dominated combined insult, showing ground truth, predictions, and absolute errors.
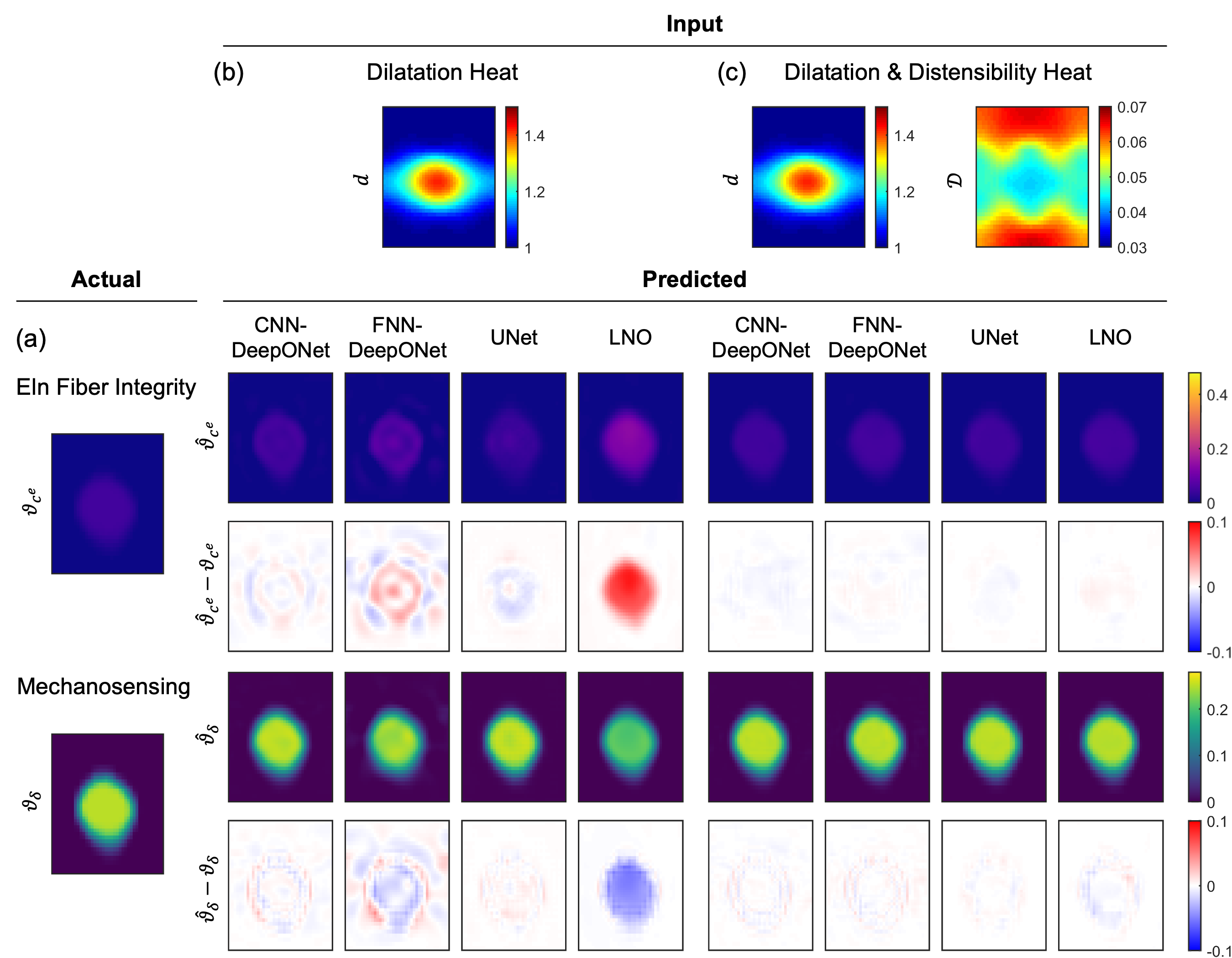
Figure 7: Predictions from all four architectures for a mechanosensing-dominated combined insult, highlighting the impact of input data format and architecture.
The choice between grayscale and heat map input formats has minimal impact on predictive accuracy for CNN-based models and LNO. FNN-DeepONet exhibits improved performance with heat maps in the presence of distensibility data but degrades in dilatation-only cases. UNet performance is robust to input format.
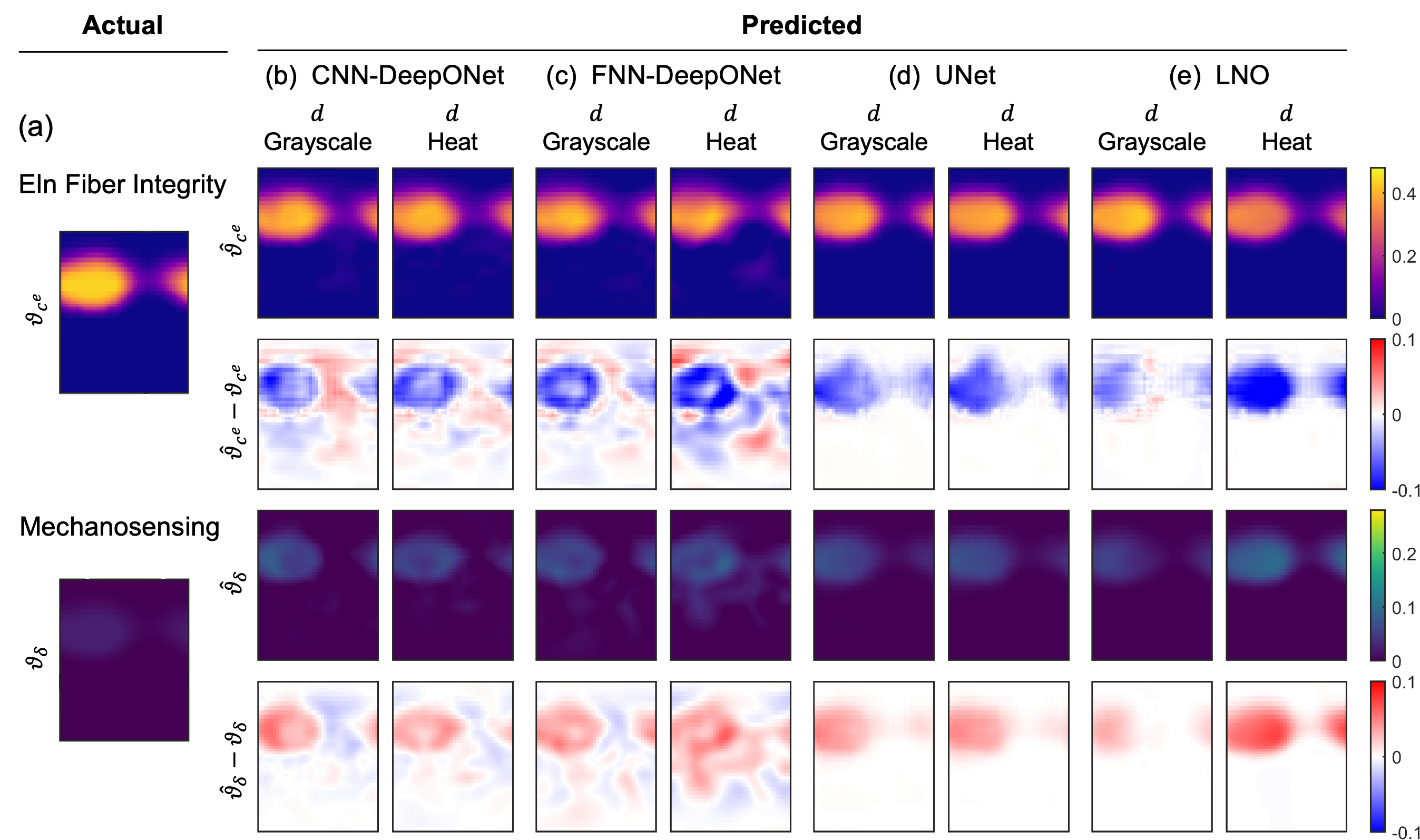
Figure 8: Effects of grayscale versus heat map data inputs on prediction accuracy across architectures.
Importance of Distensibility
The inclusion of distensibility maps is essential for disambiguating the contributions of elastic fiber integrity and mechanosensing, as these can produce similar dilatation profiles but distinct distensibility patterns. Mechanosensing-dominated insults yield higher dilatation, while elastic fiber integrity loss is associated with lower distensibility. This distinction is critical for inferring mechanical vulnerability and potential risk of dissection or rupture.
Discussion
The results establish that full-field, image-derived measurements of both dilatation and distensibility are necessary for accurate, noninvasive inference of the mechanobiological drivers of TAA. The UNet architecture is identified as the preferred model due to its superior accuracy and spatial localization capabilities, particularly in the presence of multi-contributor insults. The study demonstrates that reliance on geometric metrics alone, as in current clinical guidelines, is insufficient for mechanistic inference and risk stratification.
The synthetic data pipeline, while parameterized to murine models and limited to two insult types, provides a scalable framework for future integration of additional pathophysiological factors, patient-specific geometries, and experimental data. The approach is extensible to time-evolving datasets, where LNO may offer advantages due to its temporal modeling capabilities.
Implications and Future Directions
The findings have direct implications for the development of personalized, image-based diagnostic tools for TAA. The demonstrated necessity of distensibility measurements motivates the adoption of advanced imaging protocols capable of capturing dynamic mechanical properties. The operator learning framework is compatible with rapid, automated segmentation and analysis pipelines, enabling real-time, patient-specific risk assessment.
Future work should focus on expanding the insult space, incorporating human data, and validating the models on clinical cohorts. The integration of neural operator surrogates with high-fidelity biomechanical simulations and experimental measurements will further enhance the translational potential of this approach.
Conclusion
This study provides a rigorous, comparative evaluation of neural operator architectures for the inference of mechanobiological insult profiles in TAA from imaging-derived data. The results demonstrate that:
- Full-field measurements of both dilatation and distensibility are essential for accurate mechanistic inference.
- UNet architectures offer superior performance for spatially resolved prediction tasks in this domain.
- The operator learning framework is robust to input data format and scalable to more complex, clinically relevant scenarios.
The methodology establishes a foundation for the development of noninvasive, personalized diagnostic tools for TAA, with the potential to inform risk stratification and guide therapeutic decision-making.







