- The paper presents a novel geometric model that defines a close-packed triple helix with maximal volumetric packing (f_CP=0.744) at a 43.3° pitch angle.
- It reveals a central channel of approximately 1.06 Å that may facilitate ion transport and clarify structural ambiguities in traditional collagen models.
- Support from X-ray diffraction and NMR studies corroborates the model's potential impact on understanding biomineralization and the mechanical behavior of collagen.
The Close-Packed Triple Helix as a Structural Motif for Collagen
Introduction
The structure of collagen, the primary structural protein in animal connective tissues, has long been a subject of rigorous investigation. Traditional models, notably the 10/3 and 7/2 supercoiled helices, have been predominant, yet both exhibit significant ambiguity in structural assignments and suboptimal agreement with experimental data. In "The close-packed triple helix as a possible new structural motif for collagen" (1004.1781), Bohr and Olsen introduce a geometric analysis leading to a close-packed (CP) triple helix as the optimal packing conformation for three interwoven helical chains, proposing this as a candidate motif for collagen. This essay presents a detailed technical analysis of their geometric framework, supporting evidence from diffraction and NMR, and a critique of its implications in structural biology.
Geometric Rigour of the Close-Packed Motif
Bohr and Olsen define the close-packed triple helix by maximizing the volumetric packing fraction fV of three identical tubes arranged helically about a common axis, in accordance with hard wall geometry constraints. They analytically derive that the maximal packing is achieved at a pitch angle vCP=43.3∘, resulting in fCP=0.744, which is approximately 20% higher than that of conventional supercoiled models. The resulting topology is distinct from both the 10/3 and 7/2 motifs, as it is not supercoiled but rather a pure triple helix about a single axis.

Figure 1: Canonical topologies of triple helices including (a) supercoiled 10/3, (b) supercoiled 7/2, and (c) the CP triple helix; only the CP form achieves maximal packing among three tubes.
Numerical maximization of fV as a function of pitch angle yields the unique CP structure. Importantly, all packed triple helices possess a central longitudinal channel of nonzero radius, a feature absent in tightly packed double helices above 45∘ pitch.
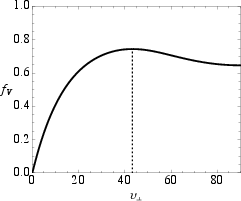
Figure 2: Packing fraction fV for triple helix; the maximum at vCP=43.3∘ defines the close-packed state.
Central Channel: Implications for Biofunctionality
The CP triple helix inherently includes a central channel of radius Ri, calculated for the canonical collagen tube diameter to be approximately $1.06$ Å. The central channel’s existence is a robust geometric consequence of optimal packing and is postulated to play a functional role—potentially as a conduit for ions or protons.
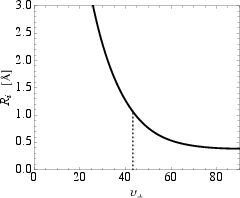
Figure 3: Minimum inner radius Ri of CP triple helix as function of pitch angle; for the collagen model, Ri=1.06 Å at vCP=43.3∘.
This architectural feature supports hypotheses regarding ion transport, proton ordering, and may be foundational for understanding physical phenomena such as the NMR magic angle effect and biomineralization processes occurring in situ within connective tissues.
Zero-Twist Structure and Molecular Mechanics
A notable mechanical attribute of the CP structure is its near-identity with the so-called zero-twist (ZT) motif—here, strain and twist decouple geometrically, minimizing strain-induced twist distortions. Quantitatively, the ZT angle vZT=42.8∘ essentially matches vCP, a unique property among possible multi-strand helices (contrast: for double helices vZT and vCP are separated by 7∘).
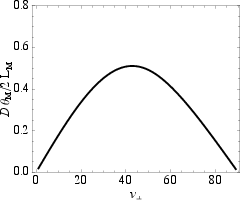
Figure 4: Normalized helical twist versus pitch angle. At vZT=42.8∘, the structure is devoid of strain-twist coupling, nearly coincident with the close-packed value.
This geometrical congruence strongly motivates the biological relevance of the CP triple helix for highly extended, force-bearing molecules like collagen, especially in tendon and bone.
Experimental Corroboration: X-ray Diffraction and NMR
Bohr and Olsen simulate X-ray scattering from idealized models, showing that only structures with a central channel produce characteristic intensity in the $1$–$2.5$ Å−1 range.
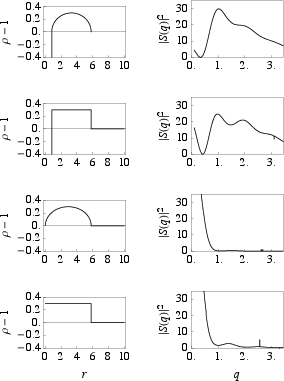
Figure 5: Calculated density and X-ray intensity profiles for tube and box models, with and without central channels; only models with a channel yield finite intensity at relevant q.
This theoretical prediction aligns with classic equatorial X-ray data from Haisa, which show features compatible with the presence of a central channel, contrary to the expectations from the conventional supercoiled collagen models.
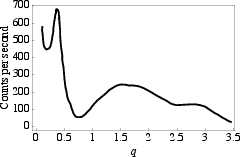
Figure 6: Experimentally measured X-ray intensity for native collagen, exhibiting a profile consistent with a channel, in agreement with the CP model calculations.
Furthermore, the NMR magic angle effect—manifest as the vanishing of dipolar splitting at ϵ=55∘—is rationalized by the existence of aligned protons within the channel, offering a direct structural-mechanical link.
Atomistic Modeling and Implications for Biomineralization
Conversion of atomistic structures from crystallographic supercoiled models (PDB: 1k6f, 2drx) to CP conformations reveals a sharp reorganization: Hydroxyproline OH side-chains arrange into two regular helical lines along the CP motif, and peptide oxygen atoms orient toward the central channel. This configuration potentially fosters a negatively charged channel wall, facilitating proton localization or ionic transport.
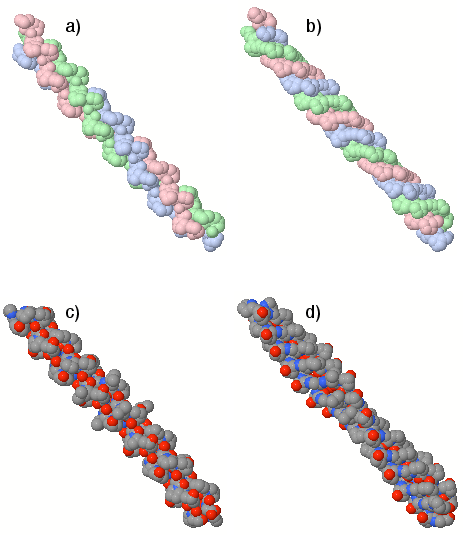
Figure 7: Comparison of atomistic models: supercoiled (a,c) and constructed CP (b,d) arrangements; note the ordered disposition of hydroxyproline OH groups and channel-facing oxygens in CP.
The implications for biomineralization are significant; the channel may provide a conduit for Ca2+ and phosphate ions, aiding uniform and spatially controlled nucleation of hydroxyapatite or other mineral phases, addressing long-standing questions regarding spatial uniformity of collagen-based mineralization.
Structural Assignment Ambiguity and CD Spectroscopy
The CP motif offers a resolution to the persistent ambiguity in collagen’s structural assignment. The failure to fit crystallographic data with low R-factors and the inadequacy of circular dichroism spectral models using standard motifs both suggest missing geometric or topological features, potentially supplied by the packed triple helix. The predicted arrangement of functional groups and channel presence may directly affect the interpretation of CD spectra, proton NMR, and other biophysical measurements.
Conclusion
The analytic derivation of the close-packed triple helix defines a unique, maximally efficient geometry for three-chain helices with explicit prediction of a central channel, a feature absent in canonical supercoiled models. Strong numerical results include the packing fraction fCP=0.744 at vCP=43.3∘, and the near-equivalence of zero-twist and close-packed geometries, an outcome not shared by other helical types. The inherent channel provides explanations for diffraction, NMR signatures, and offers a plausible mechanism for ion transport and biomineralization within collagenous tissues. The CP motif therefore demands experimental re-evaluation of collagen structure using next-generation imaging and spectroscopic techniques, and may influence biomaterials design by highlighting the functional capability of topology-driven channel formation in triple helices.






