- The paper introduces an ESP approach to evolve neural network prescriptors, achieving novel strategies for personalized insulin delivery.
- It employs a Random Forest model with an MAE of 0.5419 to accurately predict glucose deviation and validate improved stabilization outcomes.
- The integration of an interactive user interface enables dynamic prescription management, empowering informed decisions in diabetes care.
Optimizing the Design of an Artificial Pancreas to Improve Diabetes Management
The paper "Optimizing the Design of an Artificial Pancreas to Improve Diabetes Management" focuses on utilizing advanced computational models for optimizing insulin delivery systems in managing Type 1 diabetes. It introduces an Evolutionary Surrogate-assisted Prescription (ESP) approach to optimize treatment decisions for improved patient outcomes.
Introduction to Artificial Pancreas Systems
An artificial pancreas is a closed-loop system designed to automate insulin delivery to maintain glucose levels within a target range, minimizing the need for manual insulin injections (Figure 1).
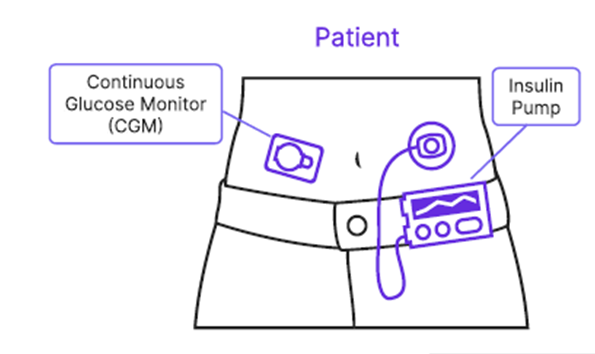
Figure 1: An artificial pancreas, i.e., a closed-loop system to manage diabetes.
The system involves continuous monitoring of blood glucose levels through a CGM and delivering insulin via a pump. The primary objective is to stabilize blood glucose levels while minimizing bolus injections. This study used a dataset from a 30-day glucose monitoring period of a single patient to train models for predicting glucose deviations and prescribing optimal carb intake, basal pumping, and bolus injections.
Evolutionary Surrogate-assisted Prescription (ESP)
ESP is introduced as a powerful technique for optimizing decision policies in real-world domains. It employs evolutionary algorithms to refine neural network-based prescriptors, leveraging surrogate models trained on historical data to predict outcomes and evaluate decision policies (Figure 2).
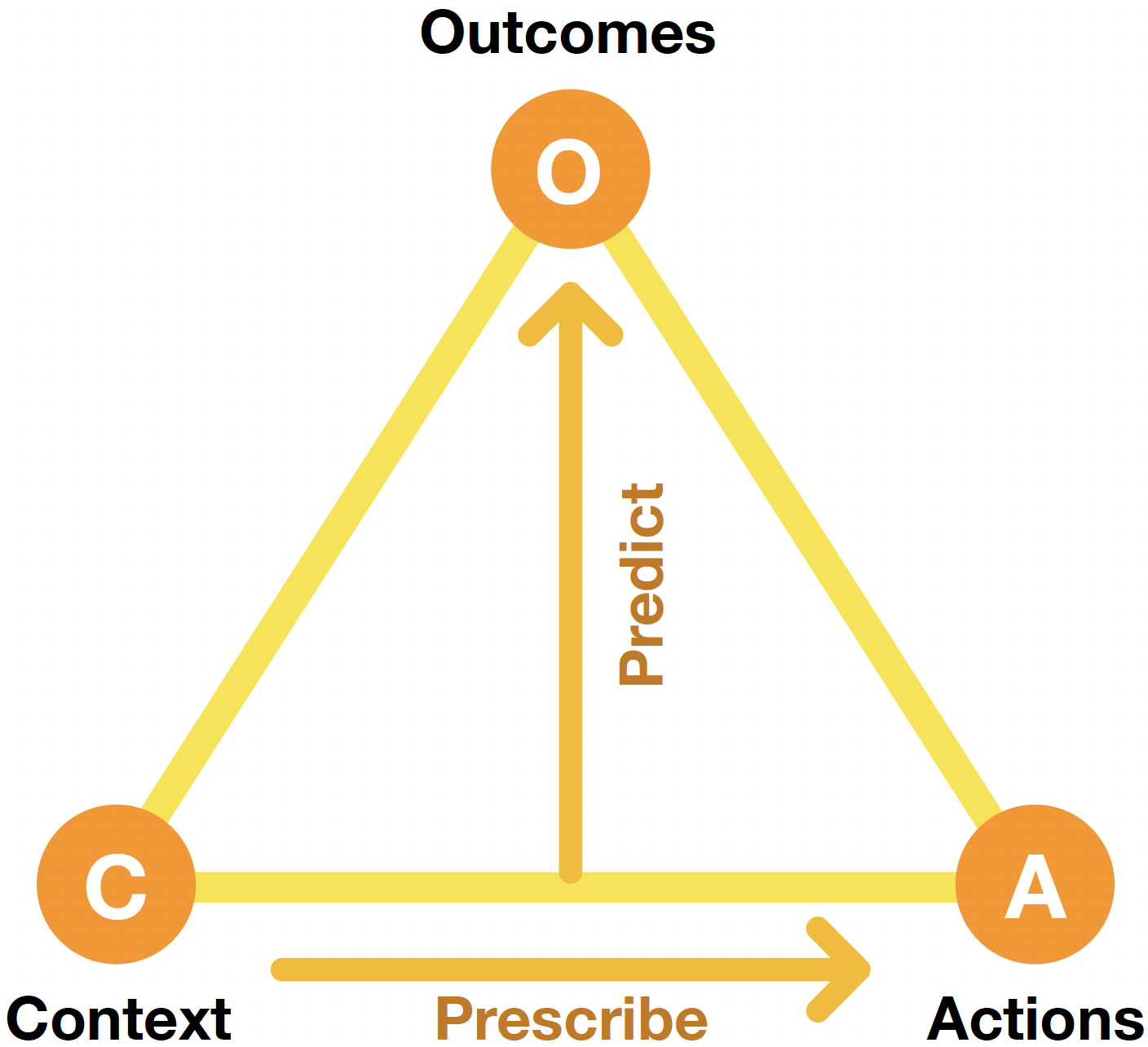
Figure 2: The ESP Decision Optimization Method.
In this context, ESP aims to discover decision strategies that lead to optimal management of blood glucose levels. Prescriptively, ESP suggests continuous values for carb intake, basal rate, and bolus dosage based on contextual data, where optimal action policies are evolved via evolutionary computation to maximize patient outcomes.
Application to Diabetes Management
Dataset and Experimental Setup
Data from the DiaTrend dataset provided input on glucose monitoring, basal rates, and insulin dosages for ESP. The dataset was aligned and aggregated into hourly samples. Two primary tasks were addressed:
- Prediction: Using a random forest model to predict blood glucose deviation based on historical data.
- Prescription: Evolving neural networks to propose action policies minimizing glucose deviation and bolus injections.
Two experiments were conducted: one constrained within the dataset's action ranges, and another exploring a broader action space to discover novel treatment strategies.
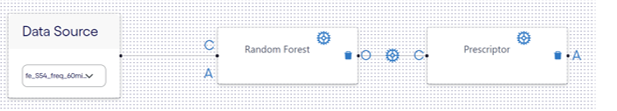
Figure 3: Building blocks for the ESP approach to managing diabetes.
Results and Analysis
The Random Forest predictor achieved an MAE of 0.5419, adequate for prescriptive model evaluation. Evolution of prescriptors demonstrated significant improvement over existing treatments, achieving superior blood glucose stabilization with potentially zero bolus injections in some scenarios (Figure 4).
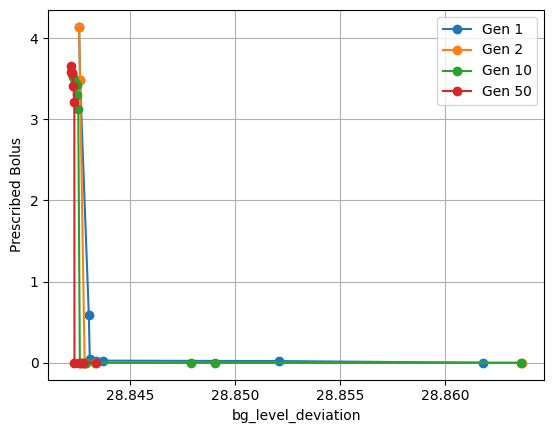
Figure 4: Progress of evolution towards better prescriptors to optimize both outcomes.
The widened action range experiment revealed enhanced outcomes and new treatment possibilities, further affirmed by comparison with original dataset outcomes (Figure 5).
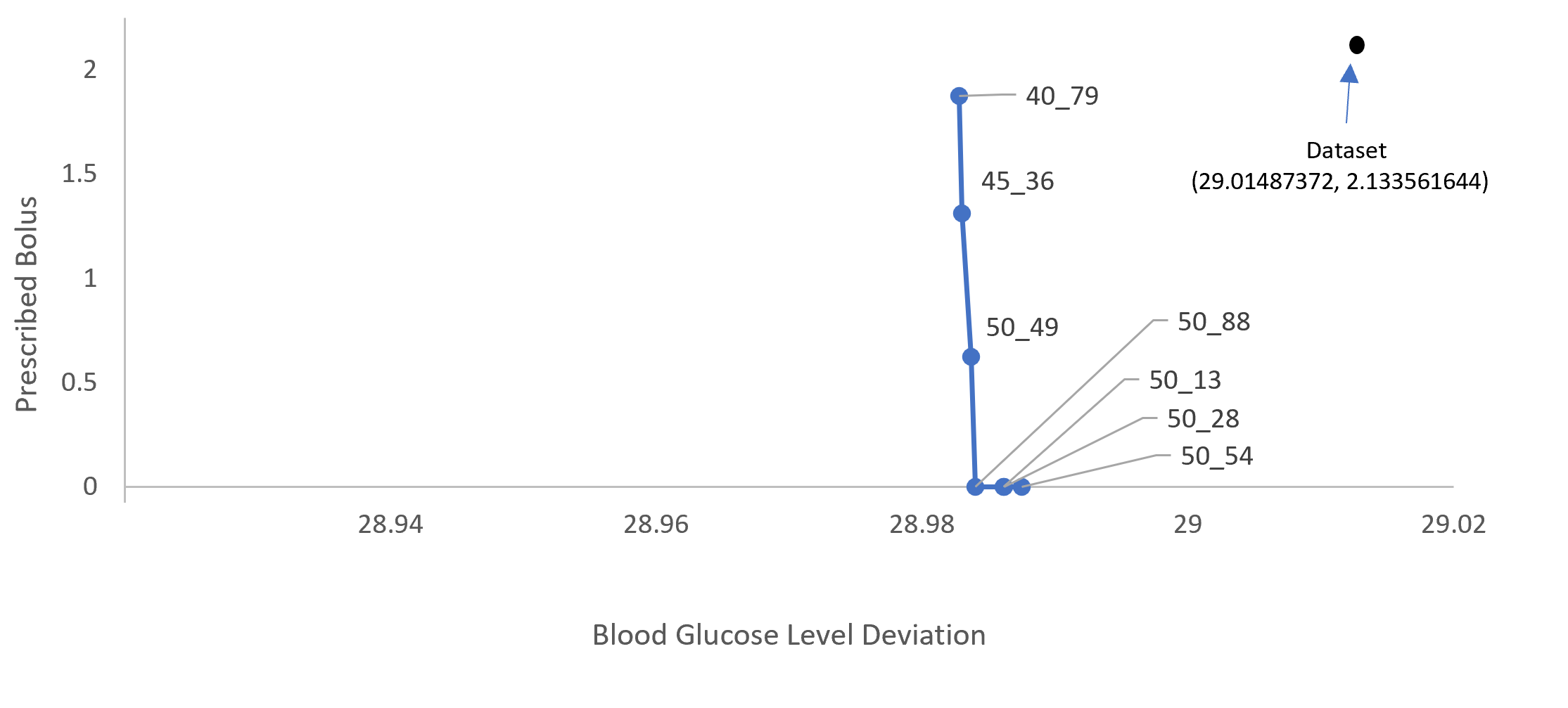
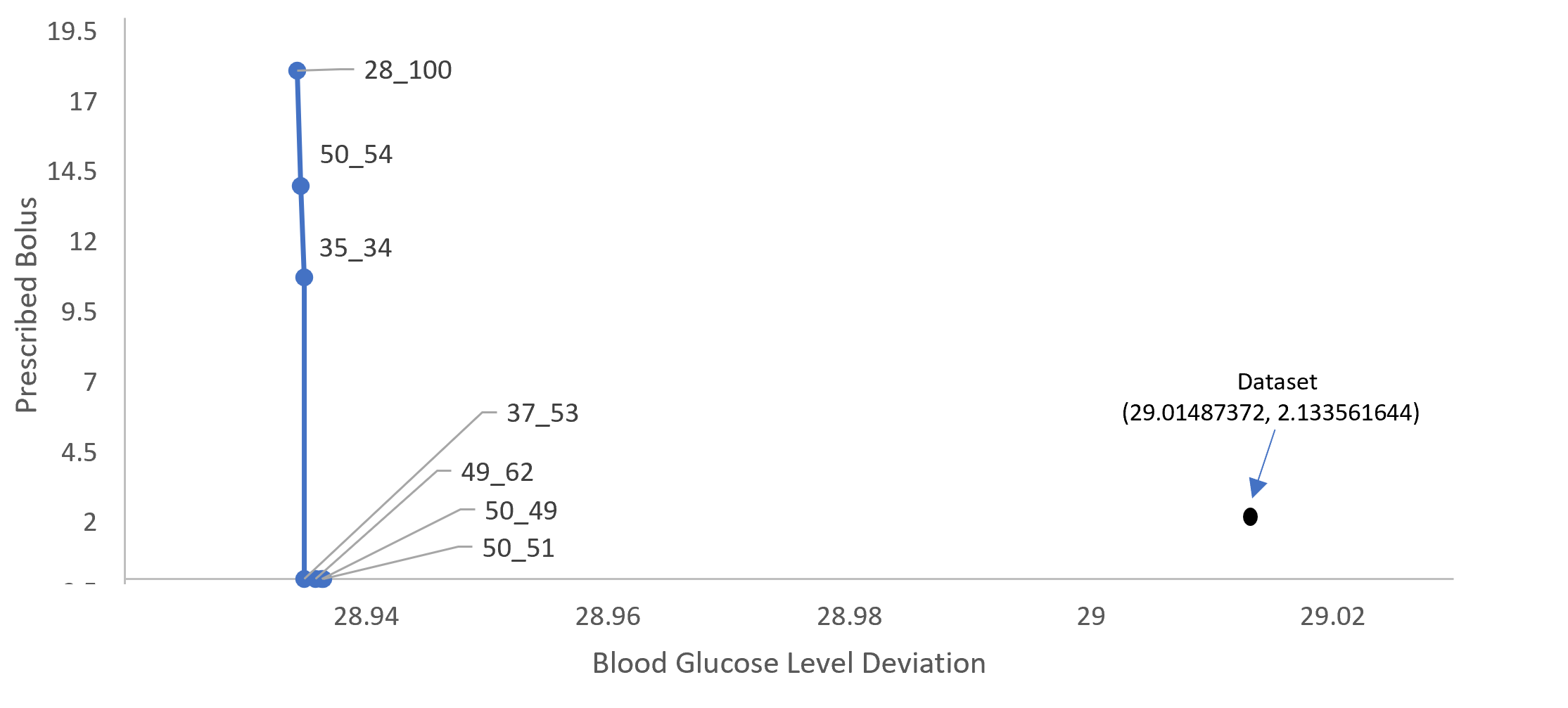
Figure 5: The final Pareto front compared to the actual outcomes in the dataset.
Interestingly, prescriptor-recommended strategies displayed novel resource utilization, such as leveraging basal rate increases over bolus injections (Figure 6), offering creative albeit short-term solutions.
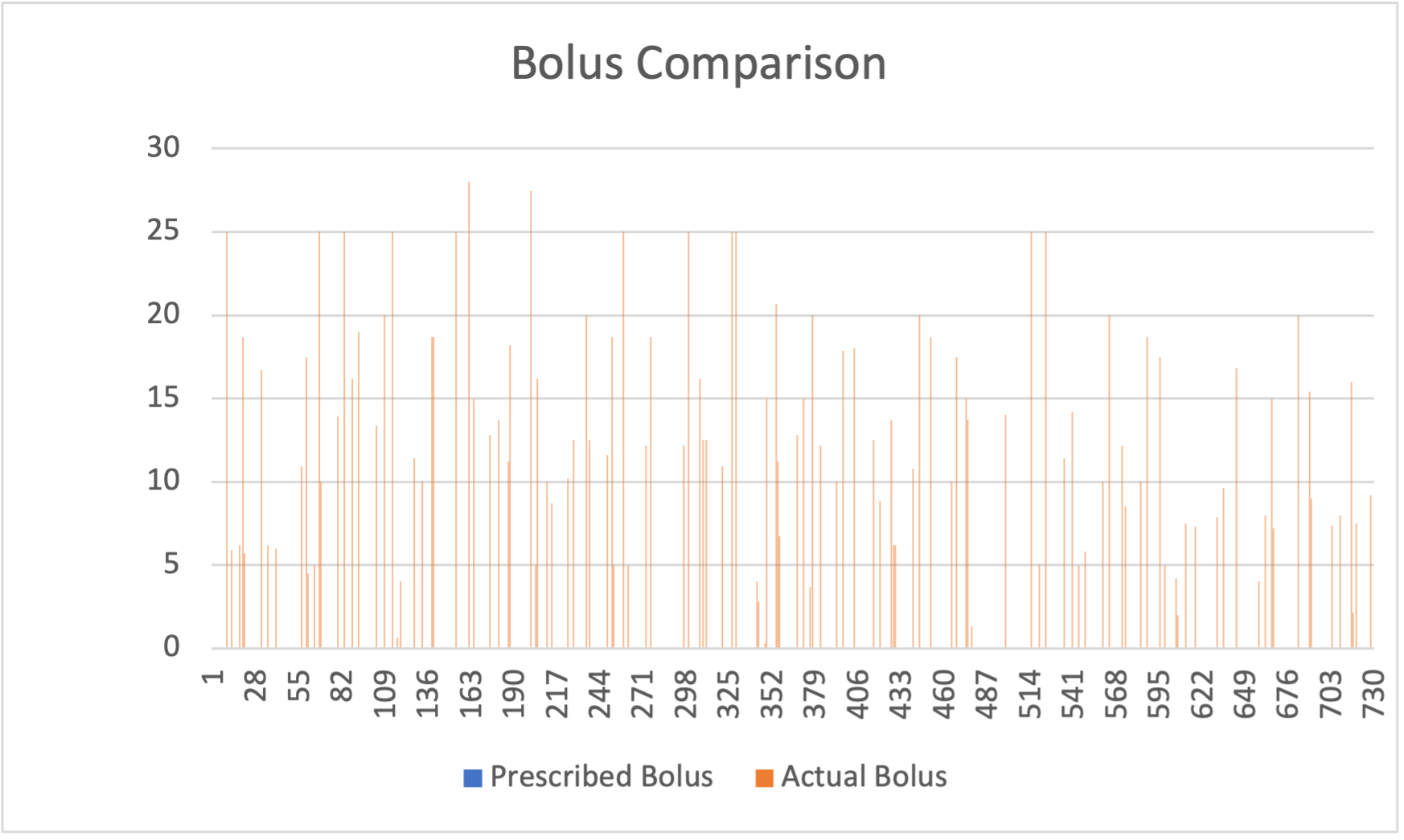
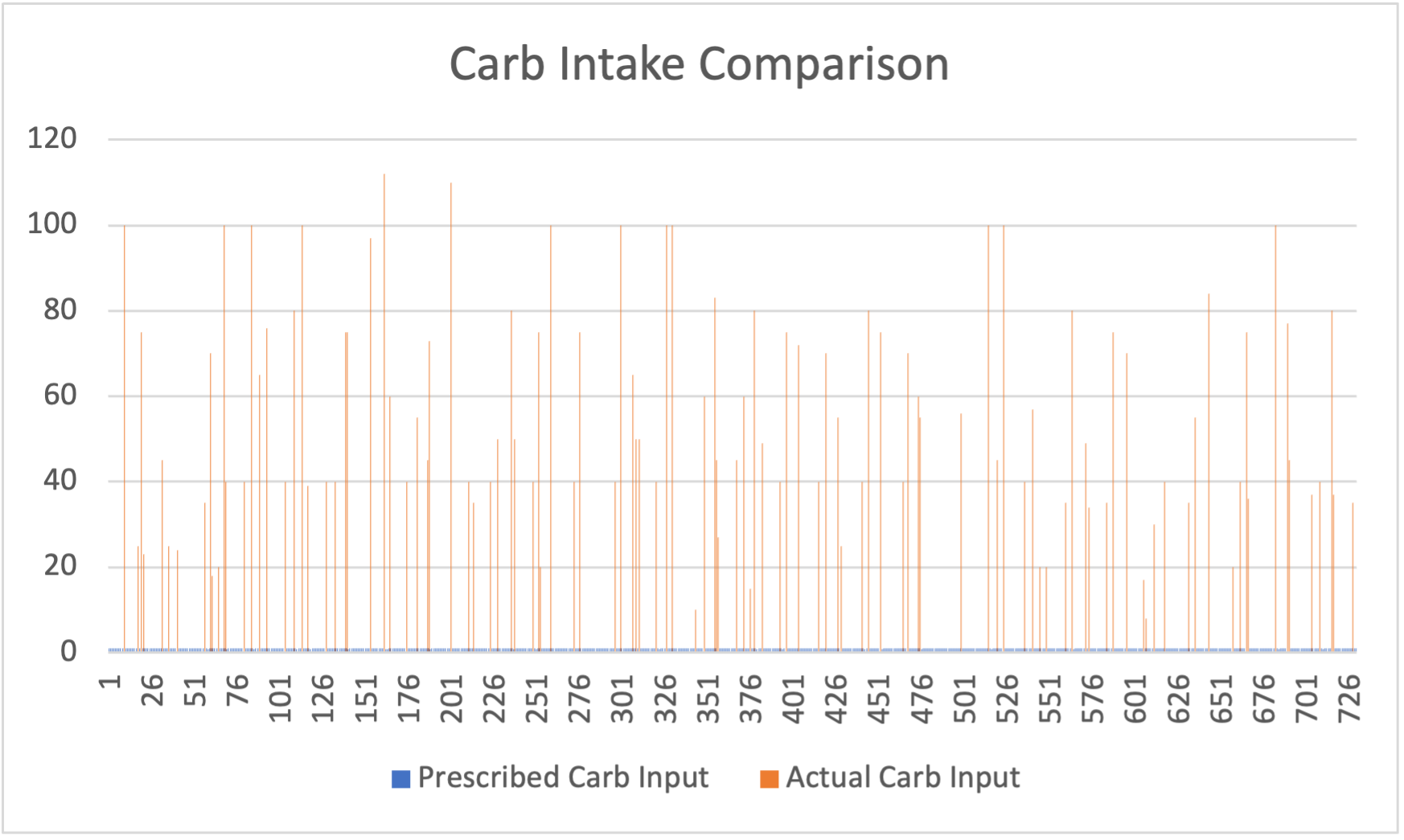
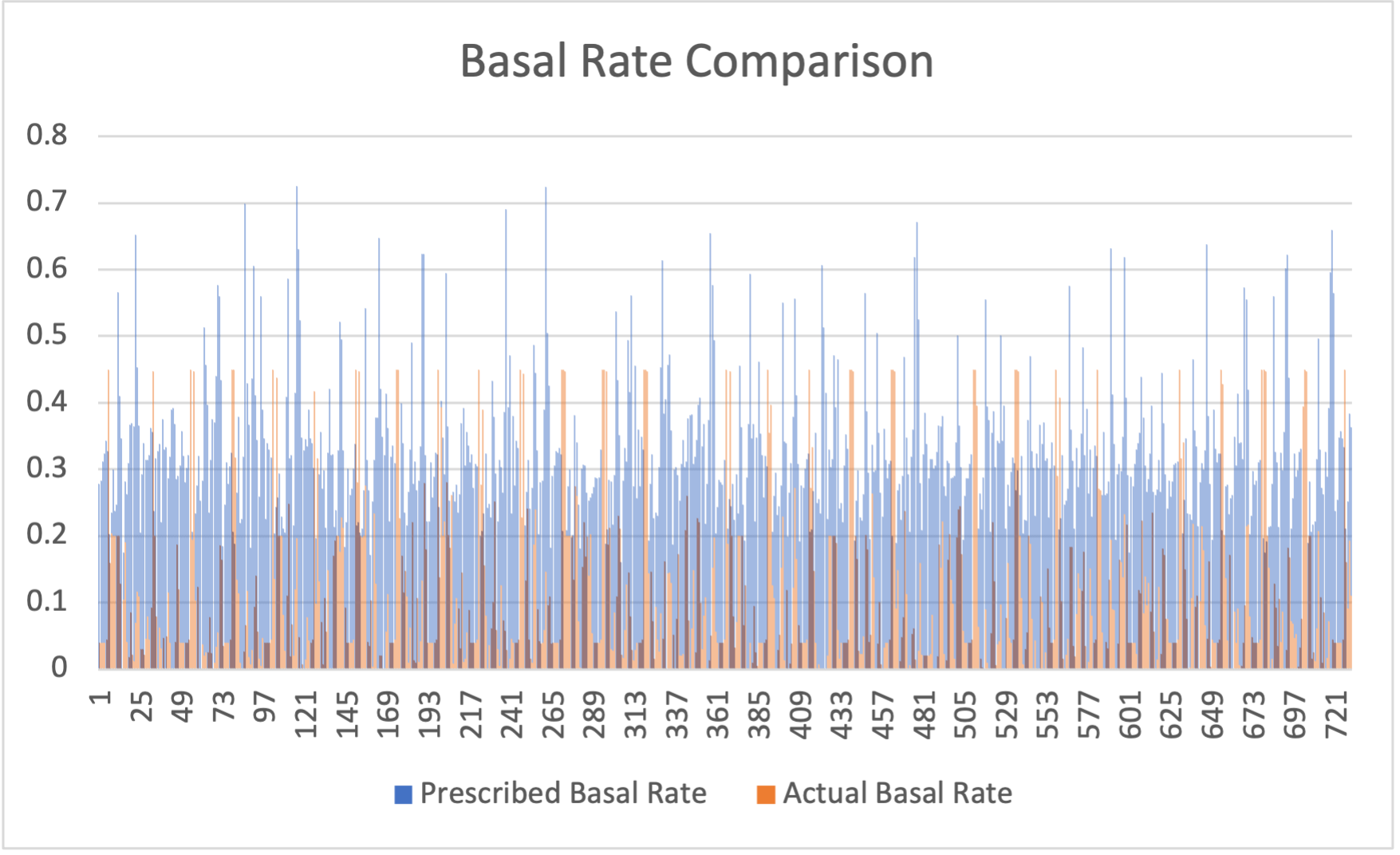
Figure 6: Comparison of the prescribed actions with the actual ones recorded in the dataset.
User Interface Integration
To facilitate real-world application, a LangChain ZeroShotReactAgent integrated into an LLM (GPT-4) interface allows interactive prescription management. The system permits users to input preferences and obtain dynamic recommendations, fostering informed patient and caregiver decisions (Figure 7).
Figure 7: The user interface for caregiver and patients during deployment.
Conclusion
The integration of ESP with diabetes treatment significantly enhances decision-making in artificial pancreas systems, proposing both incremental improvements and novel strategies for insulin management. This approach, coupled with an interactive user interface, holds potential for broad clinical adoption, enhancing quality of life for millions with diabetes. Future developments may include expanding datasets, incorporating additional biological factors, and extending model capabilities for broader patient demographics.









