- The paper presents a comprehensive computational analysis of 590 defect models, revealing that grain boundaries lower the work of adhesion and serve as preferred sites for crack initiation.
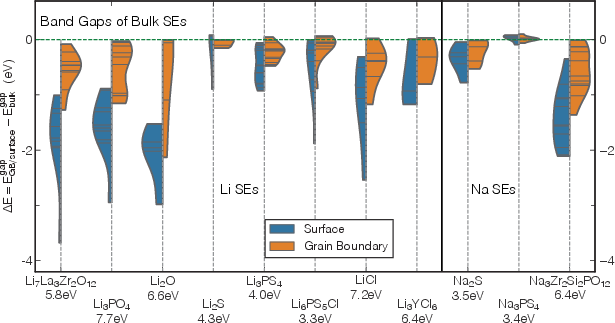
- It demonstrates that extended defects notably reduce the local electronic bandgap and introduce donor/acceptor states, potentially promoting Li/Na filament growth.
- The study highlights that synthesis conditions and microstructural design critically modulate defect energetics, influencing both mechanical stability and interfacial electronic properties.
Effects of Grain Boundaries and Surfaces on Electronic and Mechanical Properties of Solid Electrolytes
Introduction and Motivation
Polycrystalline solid electrolytes (SEs) are critical enablers for all-solid-state batteries (ASSBs) due to their potential to simultaneously achieve high energy density, safety, and stability against Li/Na metal anodes. However, the microstructure of SEs is inherently rich in extended defects—namely, grain boundaries (GBs) and surfaces—which have profound impacts on the mechanical integrity and electronic behavior of battery materials. Understanding how these defects dictate fracture initiation, ionic/electronic conduction, and interfacial stability is essential for rational design and failure mitigation in ASSBs. This study presents a comprehensive computational analysis, leveraging a library of 590 surface and GB models for 11 SE chemistries (oxides, halides, sulfides), allowing a systematic investigation of both mechanical and electronic influences of extended defects.
Construction of Grain Boundaries and Surfaces
The computational protocol began with construction of representative low-energy surfaces and twin-type GBs for each SE composition, based on experimentally reported bulk structures. Surface models were restricted to low Miller indices and constructed to be charge-neutral and non-polar, with preservation of polyanionic moieties when present. The use of Ewald energy minimization ensured physically meaningful slab and GB terminations. For GBs, symmetric twinning boundaries were prioritized, as these are established to dominate polycrystalline population at thermodynamic equilibrium. The approach enabled a robust statistical sampling of defect energetics, formation volumes, and local chemistry across a broad spectrum of realistic defect motifs.
Mechanical Properties: Cleavage, Adhesion, and Fracture
The mechanical response of SEs at extended defects was characterized by evaluating surface energies (γ), GB excess energies (σ), work of adhesion (Wad), cleavage energies (Wf), and the fracture toughness (KIc). The work of adhesion (Wad) serves as a key descriptor of the ease of intergranular separation. The analysis reveals that:
- Work of adhesion at GBs (Wad) is systematically lower than in the bulk (Wf) for most SEs, indicating grain boundaries serve as preferred fracture initiation sites.
- Oxide frameworks (e.g., LLZO, NaSICON) exhibit significantly higher Wf and Wad than phosphates, thiophosphates, and halides, reflecting their inherent lattice rigidity.
- Chloride and argyrodite SEs possess remarkably low Wad and Wf (as low as 0.13 J m−2), predicting facile cracking under stress.
- Synthesis environment (e.g., S-rich vs. S-poor conditions for argyrodites) can substantially modulate the population and strength of GBs by controlling their local excess composition and resulting defect energies.

Figure 1: Predicted work of adhesion, Wad, for representative grain boundaries in various SE chemistries, indicating high mechanical vulnerability at SE grain boundaries relative to the bulk.
Furthermore, the excess volume (Ω) accommodated at GBs, quantifying the local dilation or contraction, correlates positively with σ. High excess volume boundaries are more susceptible to population suppression under applied pressure (e.g., through sintering), pointing to a path for microstructural optimization.
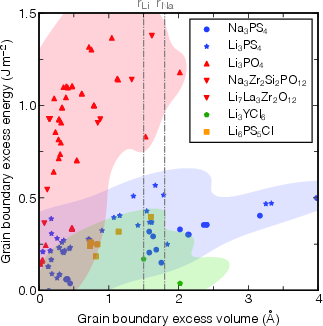
Figure 2: Relationship between excess volume Ω and GB excess energy σ across SE chemistries, demonstrating large excess volumes generally associate with thermodynamically less stable GBs.
The fracture toughness analysis establishes lower bounds for GBs and upper bounds for bulk regions. LLZO displays the highest estimated KIc, both for bulk and GBs, yet even these values are not sufficient to guarantee immunity from Li/Na intrusion when considering the stress field magnitudes arising during filament insertion. For sulfide and halide SEs, predicted KIc at GBs falls below 0.05 MPa·m1/2, pointing to intrinsic brittleness and heightened risk of catastrophic mechanical failure.
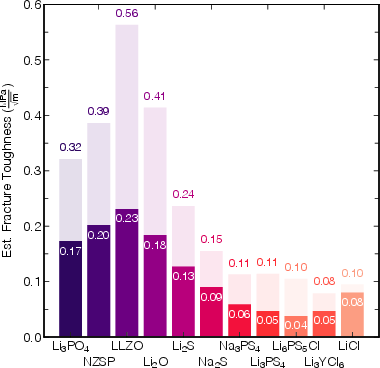
Figure 3: Estimated fracture toughness KIc for bulk and GB domains of selected SEs, with oxides showing significantly greater resistance to fracture than sulfides and halides.
Electronic Properties: Bandgap Modulation and Interfacial States
Extended defects fundamentally alter the local electronic structure of SEs. The key findings include:
Alignment of the surface energetic levels with those of Li and Na metal further elucidates the chemical stability of SEs at the metal interface. Binary compounds show large energy offsets (enhanced stability), whereas many ternary SEs, especially those with high electron affinities (e.g., certain {112} Li3PS4 surfaces), are prone to chemical reduction and instability upon contact with Li metal.
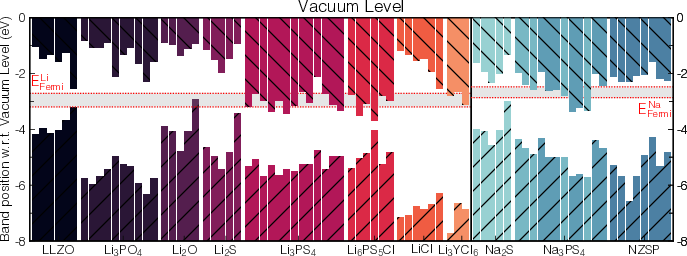
Figure 5: Alignment of SE surface electronic band edges relative to vacuum and Li/Na metal references, facilitating candidate screening for interface stability.
Discussion and Implications
The study delivers several significant insights applicable to SE design and battery engineering:
- Mechanical vulnerability at GBs dominates SE failure modes: The low Wad at GBs triggers preferential crack propagation along grain boundaries. This finding rationalizes the experimental ubiquity of intergranular fracture and motivates strategies for GB engineering or amorphization to enhance toughness.
- Synthesis and processing pathways are determinative: Thermal history, chemical environment, and densification protocol all influence the abundance and strength of GBs through their effects on both excess energy and excess volume.
- Electronic conduction pathways at defects enable filament growth: Local reductions in the electronic bandgap and the emergence of interfacial states at GBs drive heterogeneous electronic conduction, fostering Li/Na filament nucleation precisely at GBs—contradicting any supposition that bulk electronic structure alone can assess dendrite resistance.
- Microstructural design must go beyond bulk properties: For robust SE performance, both mechanical and electronic criteria must be evaluated at the microstructural (defect-inclusive) level, not merely for the pristine bulk phase.
Future developments should focus on techniques for selectively strengthening or eliminating detrimental GBs, possibly via targeted doping of GB regions, amorphization, or grain boundary passivation with inactive cations. Additionally, characterization of the dynamic response of GBs to cycling-induced stress and Li/Na accumulation is necessary for predictive lifetime modeling for practical ASSB deployment.
Conclusion
This comprehensive first-principles study of electronic and mechanical effects introduced by grain boundaries and surfaces in solid electrolytes provides a robust framework for assessing and engineering SE performance at the microstructural level. The work demonstrates that both mechanical weakness and electronic inhomogeneity at extended defects serve as primary factors in failure mechanisms such as crack initiation and Li/Na filament penetration, with profound compositional and processing dependencies. Advancing SE reliability and enabling next-generation batteries requires an integrative approach that accounts for extended defect properties in both materials selection and device design.